Clinical Evidence in Long Lesions with 48 mm XIENCE™ Stent
![]() The XIENCE Skypoint™ 48 mm Stent is designed so that interventional cardiologists (ICs) can use only one stent in long coronary lesions. Diffuse coronary disease—with long lesions—can otherwise be challenging when employing multiple stents.2
The XIENCE Skypoint™ 48 mm Stent is designed so that interventional cardiologists (ICs) can use only one stent in long coronary lesions. Diffuse coronary disease—with long lesions—can otherwise be challenging when employing multiple stents.2

Advantages in Using a Single Long Stent2
There are several advantages in using a single long stent versus several shorter stents in percutaneous coronary intervention (PCI):
- Reduces procedure time, making PCIs faster and more efficient
- Reduces radiation and fluoroscopy time
- Reduces use of contrast
Use of a single longer stent, versus 2 shorter stents, also reduces overlap. In fact, stent overlap has been reported in as many as 30% of patients undergoing PCI. Overlapping stents may also result in:
- Increased risk of stent fracture3
- Higher rates of target lesion revascularization (TLR)
XIENCE Skypoint™ Stent also offers a 4.0 mm x 48 mm stent size to help treat larger vessels where there may be diffuse disease.
Key findings from SPIRIT 48 Trial4

XIENCE™ 48 mm Stent Safety Outcomes†
Long XIENCE™ Stents continue the tradition of excellent XIENCE™ safety outcomes. XIENCE™ 48 mm Stents demonstrate impressive outcomes in long lesions:
- 100% device success (mean stented length 58 mm)5
- 0.1% definite/probable in de novo lesions >32 mm and <44 mm in length stent thrombosis (ST) at 1 year (mean stented length 39.3 mm)6


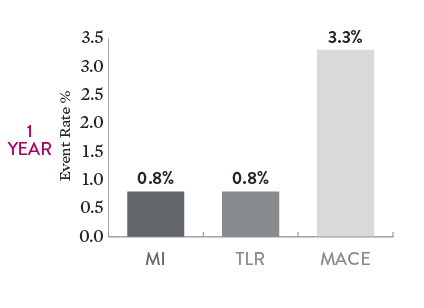
Long-Term Outcomes in Long, Complex Lesions7
When using XIENCE™ 48 mm Stents in long, complex lesions, there were low rates of myocardial infarction (MI), TLR, and major adverse cardiac events (MACE) 1 year after the index procedure. In this analysis the mean stented length was 58 mm.
Consistent Outcomes with Moderate / Long Length Stents5
Additional data supported the safety of XIENCE™ Stent in long lesions, with findings displaying consistent performance between moderate length lesions and long lesions.
| Moderate Length Lesions (> 24 mm to < 35 mm) 482 patients | Long Length Lesions (> 35 mm) 323 patients | p value | |
|---|---|---|---|
| TLF at 1 Year | 10% | 8.9% | p = 0.63 |
| Mean Lesion Length | 28.1 mm | 47.1 mm | p < 0.0001 |
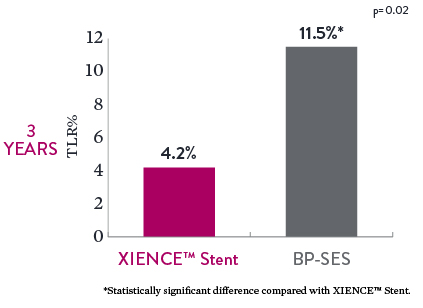
Lower Revascularization in Chronic Total Occlusions (CTO)8
Among the 3-year outcomes of the PRISON-IV trial, the XIENCE™ Stent resulted in significantly lower TLR rates of 4.2% compared to 11.5% with biodegradable polymer sirolimus-eluting stents (BP-SES). The population encompassed patients with complex CTOs.
- Total stented length with XIENCE™ Stent: 52.3 mm
- Total stented length with BP-SES: 52.4 mm
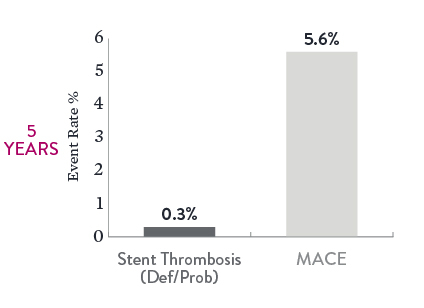
Excellent Long-Term Outcomes at 5 Years9
The 5-year data from the IVUS-XPL study reveal excellent XIENCE™ Stent outcomes in long lesion treatment guided by intravascular ultrasound (IVUS). Study results show low event rates with long lesions treated with long stents.
- Mean lesion length: 35.1 mm
- Total stented length: 39.4 mm
Note the low rates of both definite or probable ST (0.3%) and MACE (5.6%).
† XIENCE™ Stent safety outcomes include data on XIENCE Prime™ Stent & XIENCE Xpedition™ Stent.
References
- Zanchin C, et al. JACC Cardiovasc Interv. 2019;12(17):1665-1675. Serruys P, et al. N Engl J Med. 2010;363:136-146. Shiomi H, et al. JACC Cardiovasc Interv. 2019;12:637-647. Kufner S, et al. Circulation. 2019:139(3):325-333. Palmerini T, et al. Lancet. 2013;379:1393-1402. Bangalore S, et al. Circulation. 2012;125:2873-2891. Bangalore S, et al. Circ Cardiovasc Interv. 2013;6(6):378-390. Pilgrim T, et al. Lancet. 2014;384:2111-2122. Pilgrim T, et al. Lancet. 2018;392:737-746. Data on file at Abbott.
- Jurado-Román A, et al. Cardiovasc Revasc Med. 2019;20(8):681-686.
- Chinikar M, et al. Current Cardiol Rev. 2014;10:349-354.
- Park, K. et al. JSCAI 2023;10 1001.
- Tan CK, et al. Herz. 2019;44:419-424
- Hong SFJ, et al. J Am Coll Cardiol Intv. 2016;9:1438-1446.
- Bouras G, et al. Catheter Cardiovasc Interv. 2017;89(6):984-991.
- Zivelonghi C, et al. JACC Cardiovasc Interv. 2019;12(17):1747-1749. Teeuwen K, et al. JACC Cardiovasc Interv. 2017;10(2):133-143.
- Hong SJ, et al. JACC Cardiovasc Interv. 2020;13(1):62-71.
MAT-2107575 v3.0
