Don't Compromise, Take Command
The market-leading Hi-Torque Command™ 14 family of workhorse guide wires, designed for access, cross and device delivery in demanding CLTI procedures.
Proven Unique Design Elements of Hi-Torque Command™ 14 Guide Wire

Hi-Torque Command™ 14 Family is now available with enhanced design features.
Confident Crossing and Superior Device Delivery1
- New higher tip load designs for ease of lesion crossing1
- High support profiles and shorter taper length options for diverse clinical needs1
- Easy access in challenging procedures with short access-to-lesion distances*
Controlled Prolapsing and Easy Navigation
Hi-Torque Command 14 ST/MT
- Enhanced distal tip designed to form a short and tight loop that facilitates controlled prolapsing
- 1 mm 30° pre-shaped angled tip is for maneuverability1 and to facilitate navigation2 with control3 through tortuous anatomy
Superb Flexibility and Tip Durability
- Flattened tip provides flexibility to shape and reshape efficiently
- Durable nitinol tip retains shape and function throughout an entire procedure
- The Hi-Torque Command™ 14 ST/MT features a 2.5 cm radiopaque tip coil, while the Hi-Torque Command™ 14 ES comes with a 3 cm radiopaque tip coil, both designed for excellent visibility
- Hi-Torque Command™ ST/MT's pre-shaped angled tip is ready for use and to facilitate navigation and maneuverability1
Different Length and Tip Options
- Four different Tip Load Options of 2.8 g, 3.5 g, 4 g and 6 g
- Three different nitinol lengths of 10 cm, 25 cm, and 40 cm
- Four different wire support levels
- Three different wire lengths of 190 cm, 210 cm and 300 cm
- Hi-Torque Command™ 14 ST/MT Wires available in two tip-style options: pre-shaped angled tip and straight tip

* Command™ 14 ST and Command™ 14 MT
Support Profile For Hi-Torque Command™ 14 Peripheral Workhorse Guide Wires
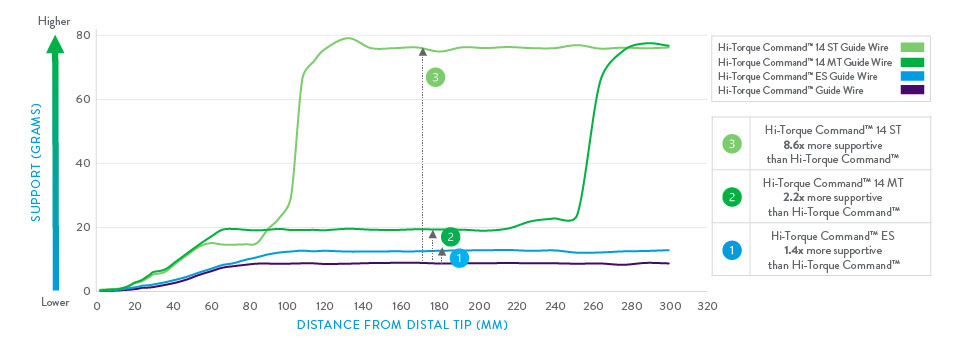
Support profile difference is calculated based on the average support levels of the wires between 150–200mm distance from the tip.
Ordering Information
| Description | Part Number | Tip Load (g) | Wire Diameter (in) | Nitinol Length | Tip Shape | Wire Length | Units Per Package |
|---|---|---|---|---|---|---|---|
| Hi-Torque Command™ 14 ST Guide Wire | 1060001 | 6 | 0.014 in | 10 cm | Straight | 210 cm | 5 |
| 1060001A | 6 | 0.014 in | 10 cm | Angled | 210 cm | 5 | |
| 1060002 | 6 | 0.014 in | 10 cm | Straight | 300 cm | 5 | |
| 1060002A | 6 | 0.014 in | 10 cm | Angled | 300 cm | 5 | |
| Hi-Torque Command™ 14 MT Guide Wire | 1060003 | 4 | 0.014 in | 25 cm | Straight | 210 cm | 5 |
| 1060003A | 4 | 0.014 in | 25 cm | Angled | 210 cm | 5 | |
| 1060004 | 4 | 0.014 in | 25 cm | Straight | 300 cm | 5 | |
| 1060004A | 4 | 0.014 in | 25 cm | Angled | 300 cm | 5 | |
| Hi-Torque Command™ ES Guide Wire | 2078174 | 3.5 | 0.014 in | 40 cm | Straight | 190 cm | 5 |
| 2078175 | 3.5 | 0.014 in | 40 cm | Straight | 300 cm | 5 | |
| Hi-Torque Command™ Guide Wire | 2078172 | 2.8 | 0.014 in | 40 cm | Straight | 190 cm | 5 |
| 2078173 | 2.8 | 0.014 in | 40 cm | Straight | 300 cm | 5 |
References
Data on file at Abbott
- Tóth, et al. "How to select a guidewire: technical features and key characteristics" Heart. 2015;101:645-652.
- Walker C. Guidewire selection for peripheral vascular interventions. Endovascular Today. 2013;5:80–3.
- Dash D. Guidewire crossing techniques in coronary chronic total occlusion intervention: A to Z. Indian Heart Journal. 2016;68:410-420.
MAT-2006300 v4.0
