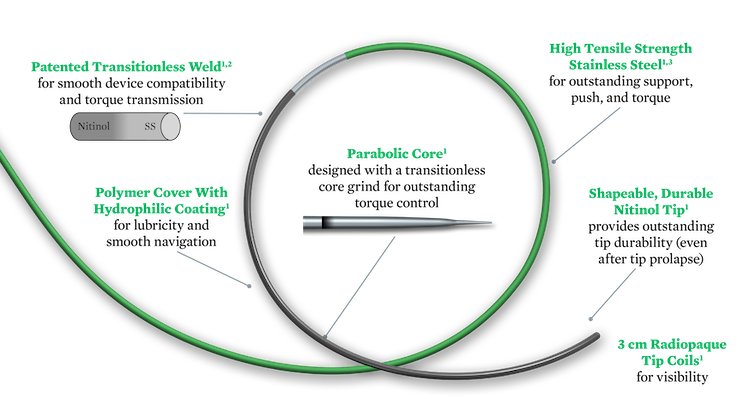
Hybrid workhorse guide wires combine the key advantages of nitinol wires and stainless steel wires.1
Take Command Of The Case
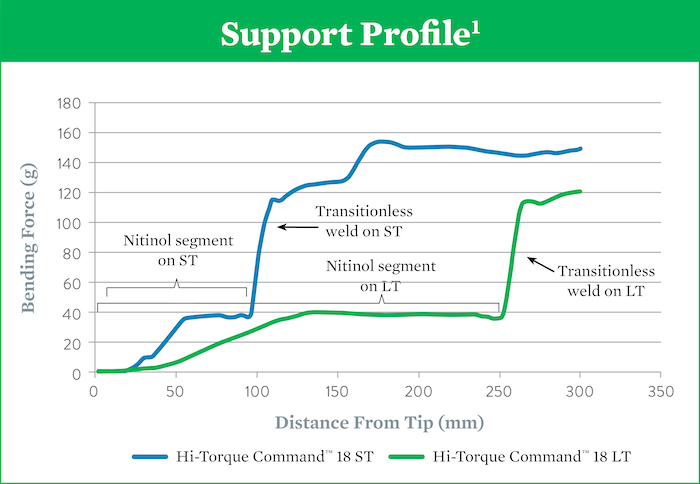
Hi-Torque Command™ 18 ST

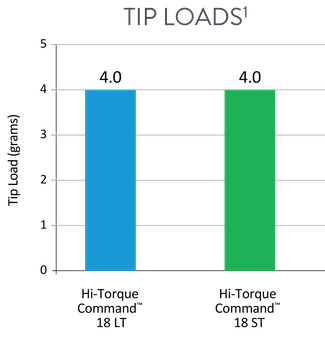
Crossing wire, with 10 cm nitinol at distal end, provides high support and a flexible tip (4g) for crossing lesions or prolapsing in a tight loop.1
Hi-Torque Command™ 18 LT

Navigation wire, with 25 cm nitinol at distal end, provides flexible support and a soft tip (4g) to effectively track through tortuous peripheral vessels.1
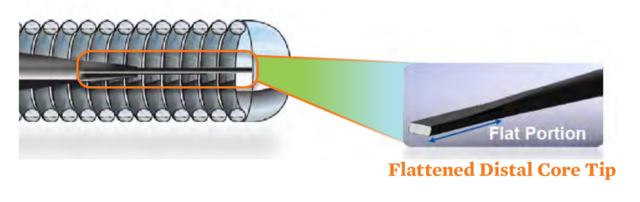
Durable and Shapeable Tip
- Shapeable flattened nitinol tip maintains its shape after prolapse and re-shaping1
- Durable tip shape helps minimize multiple guide wire usage1
Outstanding Torque Control
- Parabolic core for excellent torque control1
- Transitionless weld for torque transmission1,2
Core Material and Coating
- High tensile strength stainless steel for excellent torque, push and support1,3
- Polymer cover with hydrophilic coating for smooth navigation1
Support Profile and Tip Loads For Hi-Torque Command™ 18 Workhorse Peripheral Guide Wires
Ordering Information1
| Product | Part Number | Diameter | Length | Nitinol Length | Tip Load | Tip Shape |
|---|---|---|---|---|---|---|
| Hi-Torque Command™ 18 ST | 1013730 | 0.018 in | 210 cm | 10 cm | 4 g | Straight |
| Hi-Torque Command™ 18 ST | 1013731 | 0.018 in | 300 cm | 10 cm | 4 g | Straight |
| Hi-Torque Command™ 18 LT | 1013752 | 0.018 in | 210 cm | 25 cm | 4 g | Straight |
| Hi-Torque Command™ 18 LT | 1013753 | 0.018 in | 300 cm | 25 cm | 4 g | Straight |
Hi-Torque Command™ 18 guide wire is packaged with a torque device and guide wire introducer that can be used for tip shaping.1
References:
- Data on file at Abbott
- Toth, et al. "How to select a guidewire: technical features and key characteristics" Heart. 2015;101:645-652
- Walker. "Guidewire Selection for Peripheral Vascular Interventions". Endovascular Today 2013;80-83
MAT-2006301 v2.0
Important Safety Information
Hi-Torque Command™ 18 Guide Wire for PTA

INDICATIONS FOR USE
This Hi-Torque™ Guide Wire is intended to facilitate the placement of balloon dilatation catheters during percutaneous transluminal angioplasty (PTA), in arteries such as the femoral, popliteal and infra-popliteal arteries.This guide wire may also be used with compatible stent devices during therapeutic procedures.
The guide wire may also be used to reach and cross a target lesion, provide a pathway within the vessel structure, facilitate the substitution of one diagnostic or interventional device for another, and to distinguish the vasculature.
CONTRAINDICATIONS
Not intended for use in the coronary or cerebral vasculature.
WARNINGS
This device is not designed for use with artherectomy devices.
This device is designed and intended for ONE-TIME USE ONLY. Do not resterilize and / or reuse.
Carefully observe the instructions under “Do Not” and “Do” below. Failure to do so may result in vessel trauma, guide wire damage, guide wire tip separation, or stent damage. If resistance is observed at any time, determine the cause under fluoroscopy and take remedial action as needed. Use the most suitable guide wire for the lesion being treated.
Do Not:
- Push, auger, withdraw, or torque a guide wire that meets excessive resistance.
- Torque a guide wire if the tip becomes entrapped within the vasculature.
- Allow the guide wire tip to remain in a prolapsed condition.
- Deploy a stent such that it will entrap the wire between the vessel wall and the stent.
- Advance or withdraw the guide wire slowly.
- Use the radiopaque marker of the interventional device to confirm position.
- Examine the tip movement under fluoroscopy before manipulating, moving, or torquing the guide wire.
- Observe the wire under fluoroscopy for tip buckling, which is a sign of resistance.
- Maintain continuous flush while removing and reinserting the guide wire to prevent air from entering the catheter system. Perform exchanges slowly to prevent air entry and / or trauma.
- When reintroducing the guide wire, confirm that the interventional device tip is free within the vessel lumen and that the tip is parallel to the vessel wall.
- Use extreme caution when moving a guide wire through a non-endothelialized stent, or through stent struts, into a bifurcated vessel. Use of this technique involves additional patient risks, including the risk that the wire may become caught on the stent strut.
PRECAUTIONS
Guide wires are delicate instruments and should be handled carefully. Prior to use and when possible during the procedure, inspect the guide wire carefully for bends, kinks, or other damage. Do not use damaged guide wires. Using a damaged guide wire may result in vessel damage and / or inaccurate torque response.
Confirm the compatibility of the guide wire diameter with the interventional device before actual use.
Free movement of the guide wire within the interventional device is an important feature of a steerable guide wire system, because it gives the user valuable tactile information. Test the system for any resistance prior to use. Adjust or replace the hemostatic valve with an adjustable valve if it is found to inhibit guide wire movement.
Avoid abrasion of the hydrophilic coating.
Do not withdraw or manipulate the hydrophilic-coated wire through a metal cannula or sharp-edged object.
ADVERSE EVENTS (AEs)
Potential Adverse Events associated with use of this device may include the following but are not limited to:
- Abrupt closure
- Allergic reaction (contrast medium, drug, guide wire material)
- Amputation or limb loss
- Aneurysm or pseudoaneurysm in vessel or at vascular access site
- Angina or coronary ischemia, arrhythmia (including premature beats, bradycardia, atrial or ventricular tachycardia, atrial or ventricular fibrillation)
- Arteriovenous fistula
- Bleeding complications requiring transfusion or surgical intervention
- Critical limb ischemia
- Death
- Detachment of a system component
- Embolization (air, tissue, plaque, thrombotic material, device)
- Emergent surgery
- Fever
- Hematoma or hemorrhagic event, with or without surgical repair
- Hypotension / hypertension
- Infection
- Ischemia or infarction not covered under other AEs
- Myocardial infarction
- Occlusion
- Pain (leg, foot, back and / or insertion site)
- Perforation or rupture
- Peripheral nerve injury
- Pulmonary embolism
- Renal failure or insufficiency secondary to contrast medium (with or without treatment including dialysis)
- Restenosis
- Shock
- Stroke
- Thrombosis
- Tissue injury
- Transient ischemic attack
- Venous thromboembolism
- Vessel dissection
- Vessel spasm or recoil
- Worsening claudication
MAT-2111768 v2.0
