High Bleeding Risk Patients and DAPT Guidelines
Percutaneous Coronary Interventions (PCI) are widely used to treat complex patient populations. Post-PCI clinicians make decisions on the dual-antiplatelet therapy (DAPT) strategy.
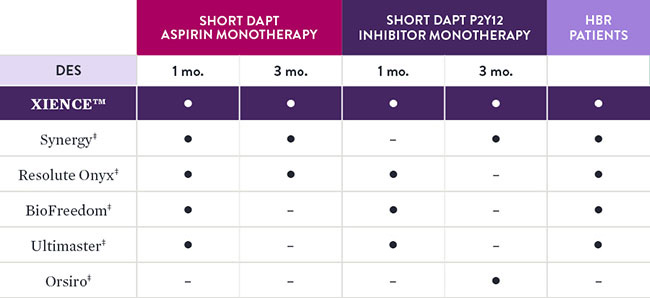
Comparison of Short DAPT Studies by Stent Type2-5
Assessing Bleeding Risk vs. Ischemic Risk in PCI Patients
It can be difficult to balance bleeding risks versus ischemic risks when determining DAPT strategy post-PCI.
Bleeding-avoidance strategies are essential to optimize patient outcomes, especially in high bleeding risk patients.

Why previous guidelines recommended longer DAPT durations — and trials omitted high bleeding risk patients
Since 2006, due to concerns about late thrombotic events, e.g. stent thrombosis (ST), an arbitrary 12-month DAPT duration was recommended.7
- High bleeding risk patients have been excluded or under-represented in clinical trials.7
- DAPT is also associated with an increased risk of bleeding, which is proportional to the duration and intensity of treatment.8
- Recent studies using the XIENCE™ Stent have been used to update the guidelines for DAPT strategy post-PCI.3,4,9
High bleeding risk criteria
In the past, clinical trials used different definitions of High Bleeding Risk.
The Academic Research Consortium (BARC) has standardized HBR definitions for patient undergoing PCI. The intention is to ensure the consistency and quality of data collection and reporting.
HBR patients were defined according to expected bleeding rates as follows:
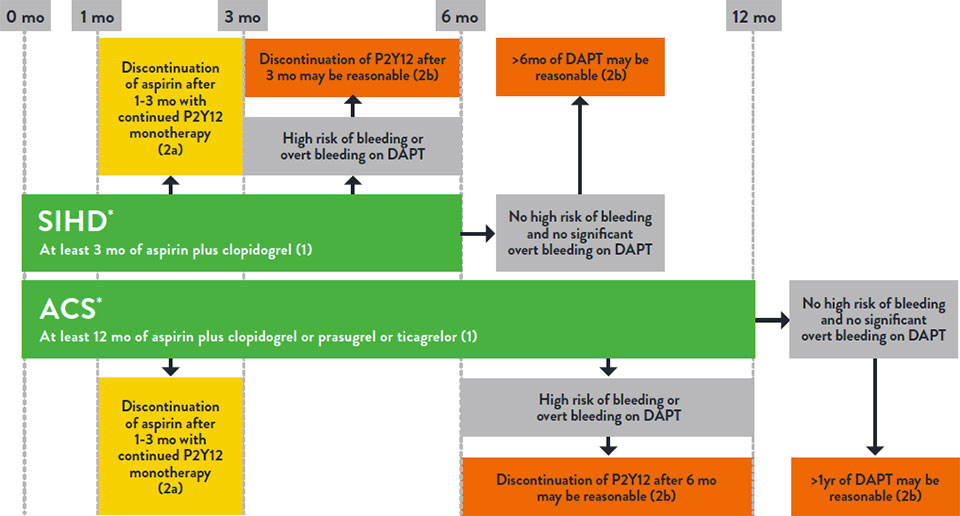
ACC/AHA/SCAI Guidelines on DAPT Strategy Post PCI10
(Adapted from Lawton J, et al 2022)
XIENCE™ Stent was a part of key studies that helped shape these guidelines and recommendations.
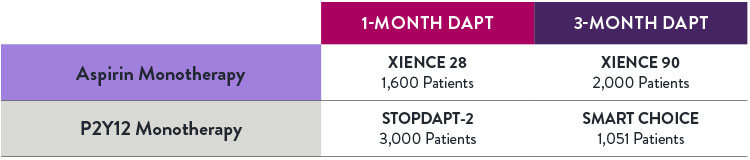
XIENCE™ Stent Body of Short DAPT Patient Evidence
A number of studies for both 1-month and 3-month DAPT have examined aspirin monotherapy and P2Y12 monotherapy—providing safety data for the various options for clinicians.3,4,9
* SIHD - Stable Ischemic Heart Disease. ACS - Acute Coronary Syndrome.
** Out of 5,769 patients in TWLIGHT STENT subanalysis, 3,014 (52.2%) had DP-EES, 1,350 (23.4%) had DP-ZES and 1,405 (24.4%) had BP-DES.
References
- XIENCE Skypoint™ Stent – Instructions for Use (IFU). Refer to IFU for additional information; Synergy‡ XD – Instructions for Use; Resolute Onyx‡ – Instructions for Use; Orsiro‡ – Instructions for Use; Promus Elite‡ – Instructions for Use. Shortest DAPT labeling as compared to all commercially available competitor DES products in the U.S. Reference: Competitor product IFUs. In HBR patients, in whom ischemic and bleeding risks must be weighed, DAPT discontinuation after 3-months or 1-month (as short as 28 days) post-PCI with XIENCE™, did not show any increase in ischemic risks.
- Mehran R, et al. JACC Cardiovasc Interv. 2021;14(17):1870-1883.
- Watanabe H, et al. JAMA. 2019;321(24):2414-2427.
- Hahn J, et al. JAMA. 2019;321(24):2428-2437.
- Varenne O, et al. Lancet. 2018.391:41-50 – SENIOR. Kirtane A, et al. TCT 2019 – EVOLVE Short DAPT. Kedhi E, et al. PCR eCourse 2020 – Onyx ONE. Postma W, et al. Catheter Cardiovasc Interv. 2020;95:706-710 – DAPT-STEMI. Valgimigli M, et al. N Engl J Med. 2021;10.1056 – MASTER DAPT.
- Capodanno D, et al. J Am Coll Cardiol. 2020;76:1468-1483.
- Urban P, et al. Eur Heart J. 2019; 40(31):2632-2653.
- Palmerini T, et al. Eur Heart J. 2017;38:1034-1043.
- Mehran R, et al. N Engl J Med. 2019;381:2032-42.
- Lawton JS, et al. J Am Coll Cardiol. 2022;79(2):e21-e129.
- Dangas G, et al. EuroIntervention. 2022;17(16):1330-1339.
MAT-2504801 v1.0
XIENCE Skypoint™, XIENCE Sierra™, XIENCE Alpine™ (XIENCE™ Family) Everolimus Eluting Coronary Stent Systems

Indications
Applies to XIENCE Skypoint™ Stent Systems:
- Indicated for improving coronary artery luminal diameter in patients, including those with diabetes mellitus, with symptomatic heart disease due to de novo native coronary artery lesions ≤ 44 mm in length with reference vessel diameters of ≥ 2.25 mm to ≤ 5.25 mm and for high bleeding risk patients with coronary arteries lesions ≤ 32 mm in length with a reference vessel diameter of ≥ 2.25 mm and ≤ 5.25 mm.
- Treating de novo chronic total coronary occlusions.
Applies to XIENCE Sierra™ and XIENCE Alpine™ Stent Systems:
- Indicated for improving coronary artery luminal diameter in patients, including those at high risk for bleeding and those with diabetes mellitus, with symptomatic heart disease due to de novo native coronary artery lesions (length ≤ 32 mm) with reference vessel diameters of ≥ 2.25 mm to ≤ 4.25 mm.
- Treating de novo chronic total coronary occlusions.
Contraindications
The XIENCE Skypoint™, XIENCE Sierra™ and XIENCE Alpine™ Stent Systems are contraindicated for use in:
- Patients who cannot tolerate, including allergy or hypersensitivity to, procedural anticoagulation or the post-procedural antiplatelet regimen.
- Patients with hypersensitivity or contraindication to everolimus or structurally-related compounds, or known hypersensitivity to stent components (cobalt, chromium, nickel, tungsten, methacrylic polymer, fluoropolymer), or with contrast hypersensitivity.
Warnings
- Each stent and the delivery system are for single use only. Do not reuse, reprocess, or resterilize. Note the product “Use by” date on the package. Reuse, reprocessing, or resterilization may compromise the structural integrity of the device and / or delivery system and / or lead to device failure, which may result in patient injury, illness, or death. Reuse, reprocessing, or resterilization may also create a risk of contamination of the device and / or cause patient infection or cross-infection, including, but not limited to, the transmission of infectious disease(s) from one patient to another. Contamination of the device and / or delivery system may lead to injury, illness, or death of the patient.
- It is not recommended to treat patients having a lesion that prevents complete inflation of an angioplasty balloon.
- Antiplatelet therapy should be administered post-procedure.
- This product should not be used in patients who are not likely to comply with the recommended antiplatelet therapy.
- Judicious selection of patients is necessary, since the use of this device carries the associated risk of stent thrombosis, vascular complications, and/or bleeding events.
- The XIENCE Skypoint™, XIENCE Sierra™ and XIENCE Alpine™ Stent Systems are coated with an everolimus and polymer coating at the full implant stent length. The distal and intermediate portions of the device, the tip, and tapers of the balloon are coated with HYDROCOAT™ Hydrophilic Coating.
Failure to abide by the warnings in this labeling might result in damage to the device coating, which may necessitate intervention or result in serious adverse events.
Precautions
- Implantation of the stent should be performed only by the physicians who have received appropriate training.
- Stent placement should be performed at centers where emergency coronary artery bypass graft surgery (CABG) can be readily performed.
- When the XIENCE Skypoint™, XIENCE Sierra™ and XIENCE Alpine™ Stent Systems are used outside the specified Indications for Use, patient outcomes may differ from the results observed in the SPIRIT family of clinical trials. Compared to use within the specified indications for use, the use of the XIENCE Skypoint™, XIENCE Sierra™ and XIENCE Alpine™ Stent Systems in patients and lesions outside of the labeled indications, including more tortuous anatomy, may have an increased risk of adverse events, including stent thrombosis, stent embolization, MI, or death.
- The extent of the patient’s exposure to drug and polymer is directly related to the number of stents implanted. See Instructions for Use for current data on multiple stent implantation.
- Safety and effectiveness of the XIENCE Skypoint™, XIENCE Sierra™ and XIENCE Alpine™ Stent Systems have not been established for subject populations with the following clinical settings:
- Patients with prior brachytherapy of the target lesion or the use of brachytherapy for treated site restenosis.
- Conjunctive use of the XIENCE Skypoint™, XIENCE Sierra™ and XIENCE Alpine™ Stent Systems with either mechanical atherectomy devices or laser angioplasty catheters.
- Women who are pregnant or lactating, men intending to father children, pediatric.
- Unresolved vessel thrombus at the lesion site, coronary artery reference vessel diameters < 2.25 mm or > 4.25 mm, for XIENCE Skypoint™ < 2.25 mm or > 5.25 mm, or lesion lengths >44 mm, lesions located in saphenous vein grafts, lesions located in unprotected left main coronary artery, ostial lesions, or lesions located at a bifurcation or previously stented lesions, diffuse disease or poor flow (TIMI < 1) distal to the identified lesions, excessive tortuosity proximal to or within the lesion, recent Acute Myocardial Infarction (AMI) or evidence of thrombus in target vessel, multivessel disease, and in-stent restenosis.
- Formal drug interaction studies have not been performed with the XIENCE Skypoint™, XIENCE Sierra™ or XIENCE Alpine™ Stent Systems because of limited exposure to everolimus eluted from XIENCE Skypoint™, XIENCE Sierra™ and XIENCE Alpine™ Stent Systems.
- Everolimus, the active ingredient in the stents, is an immunosuppressive agent. Consideration should be given to patients taking other immunosuppressive agents or who are at risk for immune suppression.
- Oral everolimus use in renal transplant and advanced renal cell carcinoma patients was associated with increased serum cholesterol and triglyceride levels, which in some cases required treatment.
- Nonclinical testing has demonstrated that the XIENCE Sierra™ and XIENCE Alpine™ Stent Systems in single and in overlapped configurations up to 71 mm, for XIENCE Skypoint™ 91 mm in length, are MR Conditional. See Instructions for Use for detailed scanning conditions
Potential Adverse Events
Adverse events that may be associated with PCI treatment procedures and the use of a stent in native coronary arteries include, but are not limited to, the following:
- Allergic reaction or hypersensitivity to latex, contrast agent anesthesia, device materials, and drug reactions to everolimus, anticoagulation, or antiplatelet drugs
- Vascular access complications which may require transfusion or vessel repair, including: Catheter site reactions, Bleeding, Arteriovenous fistula; pseudoaneurysm, aneurysm, dissection, perforation/rupture, Embolism, Peripheral nerve injury, Peripheral ischemia
- Coronary artery complications which may require additional intervention, including: Total occlusion or abrupt closure, Arteriovenous fistula, pseudoaneurysm, aneurysm, dissection. Perforation/rupture, Tissue prolapse/plaque shift, Embolism, Coronary or stent thrombosis, Stenosis or restenosis
- Pericardial complications which may require additional intervention, including: Cardiac tamponade, Pericardial effusion, Pericarditis.
- Cardiac arrhythmias
- Cardiac ischemic conditions (including myocardial ischemia, myocardial infarction (including acute), coronary artery spasm, and unstable or stable angina pectoris)
- Stroke/Cerebrovascular Accident (CVA) and Transient Ischemic Attack (TIA)
- System organ failures: Cardio-respiratory arrest, Cardiac failure, Cardiopulmonary failure, Renal Insufficiency/failure, Shock
- Bleeding
- Blood cell disorders
- Hypotension and/or hypertension
- Infection
- Nausea and vomiting
- Palpitations
- Dizziness
- Syncope
- Chest Pain
- Fever
- Pain
- Death
The risks described below include the anticipated adverse events relevant for the cardiac population referenced in the contraindications, warnings and precaution sections of the everolimus labels / SmPCs and / or observed at incidences ≥ 10% in clinical trials with oral everolimus for different indications. Please refer to the drug SmPCs and labels for more detailed information and less frequent adverse events.
- Abdominal pain
- Anemia
- Angioedema
- Arterial Thrombotic Events
- Bleeding and coagulopathy
- Constipation
- Cough
- Diabetes mellitus
- Diarrhea
- Dyspnea
- Embryo-fetal toxicity
- Erythema
- Erythroderma
- Headache
- Hepatic artery thrombosis
- Hepatic disorders
- Hypersensitivity to everolimus active substance, or to other rapamycin derivates
- Hypertension
- Infection (bacterial, fungal, viral or protozoan infections, including infections with opportunistic pathogens). Polyoma virus-associated nephropathy (PVAN), JC virus-associated progressive multiple leukoencephalopathy (PML), fatal infections and sepsis have been reported in patients treated with oral everolimus
- Kidney arterial and venous thrombosis
- Laboratory test alterations
- Lymphoma and skin cancer
- Male infertility
- Menstrual irregularities
- Nausea
- Nephrotoxicity
- Non-infectious pneumonitis
- Oral ulcerations
- Pain
- Pancreatitis
- Pericardial effusion
- Peripheral edema
- Pleural effusion
- Pneumonia
- Pyrexia
- Rash
- Renal Failure
- Upper respiratory tract infection
- Urinary tract infection
- Venous thromboembolism
- Vomiting
- Wound healing complications
There may be other potential adverse events that are unforeseen at this time.
MAT-2100879 v7.0