Supera™ Stent Lesion Preparation
Per the Instructions for Use, physicians should prepare the vessel using standard angioplasty technique; balloon size should be greater than or equal to the stent diameter. Refer to the IFU for additional information.
| Labeled Stent Diameter | Reference Vessel Diameter | Nominal Stent Length (mm) | Catheter Outer Sheath Diameter |
|---|---|---|---|
| 4.5 mm | 4.0 - 4.5 mm | 20, 30, 40, 60, 80, 100, 120, 150 | 6F 2.06 0.081 inches |
| 5.0 mm* | 4.5 - 5.0 mm | 20**, 30**, 40, 60, 80, 100, 120 | |
| 5.5 mm | 5.0 - 5.5 mm | 20, 30, 40, 60, 80, 100, 120, 150, 180, 200 | |
| 6.0 mm* | 5.5 - 6.0 mm | 20**, 30**, 40, 60, 80, 100, 120, 150 | |
| 6.5 mm | 6.0 - 6.5 mm | 20, 30, 40, 60, 80, 100, 120, 150, 180, 200 | |
| 7.0 mm* | 6.5 - 7.0 mm | 20**, 30**, 40, 60, 80, 100 | |
| 7.5 mm | 7.0 - 7.5 mm | 20, 30, 40, 60, 80, 100 |
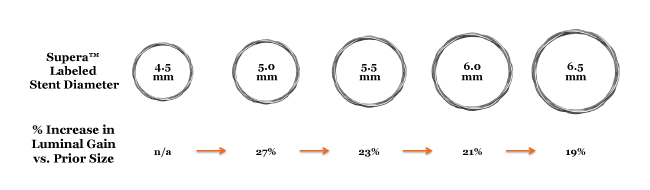
Guidelines for Stent Sizing
The illustration below shows that every 0.5 mm increase provides an estimated 19% to 27% growth in luminal gain (cross-sectional area).
The image below is a percutaneous transluminal angioplasty (PTA) balloon inflation example for a 5.5 mm Supera™ Stent.

Precaution: The vessel/duct after pre-dilatation should be at least the size of the stent diameter. If recommended, vessel/duct diameter cannot be gained, optimal stent deployment may not be achieved and revised stent sizing should be considered.
Supera™ Peripheral Stent System Instructions for Use (IFU). Refer to IFU for additional information.
Supera™ Resources
MAT-2109825 v2.0
Supera™ Peripheral Stent System

Indications
The Supera™ Peripheral Stent System is indicated to improve luminal diameter in the treatment of patients with symptomatic de novo or restenotic native lesions or occlusions of the superficial femoral artery (SFA) and / or proximal popliteal artery with reference vessel diameters of 4.0 to 7.5 mm, and lesion lengths up to 140 mm.
Contraindications
The Supera™ Peripheral Stent System is contraindicated in:
- Patients who are judged to have a lesion that prevents complete inflation of an angioplasty balloon or proper placement of the stent or stent delivery system.
- Patients who cannot receive antiplatelet or anticoagulation therapy. Based on in vivo thrombogenicity testing, the device should not be used in patients who cannot be anticoagulated as there may be some thrombus formation in the absence of anticoagulation.
Warnings
- This device is intended for single-use only. Do not reuse. Do not resterilize. Do not use if the package is opened or damaged.
- Use this device prior to the “Use By” date as specified on the device package label. Store in a dry, dark, cool place.
- DO NOT use if it is suspected that the sterility of the device has been compromised.
- Persons with known hypersensitivities to Nitinol and / or its components (e.g. nickel-titanium) may suffer an allergic reaction to this implant.
- Administer appropriate antiplatelet therapy pre- and post-procedure.
- Careful attention should be paid when sizing and deploying the stent to prevent stent elongation. In the SUPERB clinical study, stent elongation was associated with a decrease in patency at 12 months.
Precautions
The Supera™ Peripheral Stent System should only be used by physicians and medical personnel trained in vascular interventional techniques and trained on the use of this device.
- The long-term safety and effectiveness of the Supera™ Peripheral Stent System has not been established beyond three years.
- The safety and effectiveness of the Supera™ Peripheral Stent System has not been established in patients who:
- are less than 18 years old
- are pregnant or lactating
- have in-stent restenosis of the target lesion
- have known hypersensitivity to any component of the stent system (e.g., nickel)
- cannot tolerate contrast media and cannot be pre-treated
- have uncontrolled hypercoagulability and / or another coagulopathy
- This device is not designed for use with contrast media injection systems or power injection systems.
- The flexible design of the Supera™ stent may result in variation in the deployed stent length.
Magnetic Resonance Imaging (MRI) Safety Information
Nonclinical testing has demonstrated that the Supera™ stent, in single and in overlapped configurations up to 250 mm in length, is MR Conditional. A patient with this device can be safely scanned in an MR system meeting the following conditions:
- Static magnetic field of 1.5 or 3.0 Tesla
- Maximum spatial gradient magnetic field of 2,500 Gauss/cm (25 T/m)
- Maximum MR whole-body-averaged specific absorption rate (SAR) of
- 2 W/kg for landmarks (i.e. center of RF coil) above the umbilicus
- 1 W/kg for landmarks below the umbilicus and above the mid-thigh
- 0.5 W/kg for landmarks below the mid-thigh
Under the scan conditions defined above, the Supera™ stent is expected to produce a maximum temperature rise of 7.6 °C after 15 minutes of continuous scanning.
In nonclinical testing, the image artifact caused by the device extends approximately 2 cm from the Supera™ stent when imaged with a gradient echo or spin echo sequence and a 3T MRI system.
Potential Adverse Events
Potential adverse events include, but are not limited to:
- Abrupt closure
- Allergic reaction (contrast medium; drug; stent material)
- Amputation or limb loss
- Aneurysm or pseudoaneurysm in vessel or at vascular access site
- Angina or coronary ischemia
- Arrhythmia (including premature beats, bradycardia, atrial or ventricular tachycardia, atrial or ventricular fibrillation)
- Arteriovenous fistula
- Bleeding complications requiring transfusion or surgical intervention
- Death
- Detachment of a system component or implantation in an unintended site
- Embolization, arterial or other (e.g. air, tissue, plaque, thrombotic material, or stent)
- Emergent surgery
- Fever
- Hematoma or hemorrhagic event, with or without surgical repair
- Hyperperfusion syndrome
- Hypertension / Hypotension
- Infection
- Myocardial infarction
- Pain (leg, foot, and/or insertion site)
- Partial stent deployment
- Peripheral nerve injury
- Pulmonary embolism
- Renal failure or insufficiency
- Restenosis of vessel in stented segment
- Shock
- Stent malapposition or migration, which may require emergency surgery to remove stent
- Stent strut fracture
- Thrombosis or occlusion
- Stroke
- Transient ischemic attack
- Venous thromboembolism
- Vessel dissection, perforation or rupture
- Vessel spasm or recoil
- Worsening claudication or rest pain
MAT-2103597 v3.0
