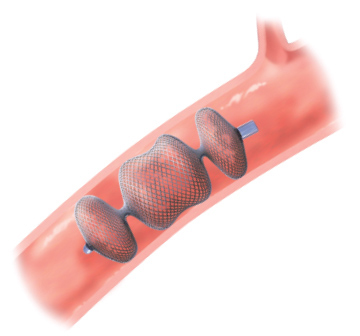
Rapid embolization1 and can adjust to variable landing zones2
- Multi-layered mesh designed to increase density and flow disturbance3
- Six planes of cross sectional coverage3 resulting in rapid occlusion1
- Select a device that is ~30-50% larger in diameter than the target vessel diameter3

Device Description3:
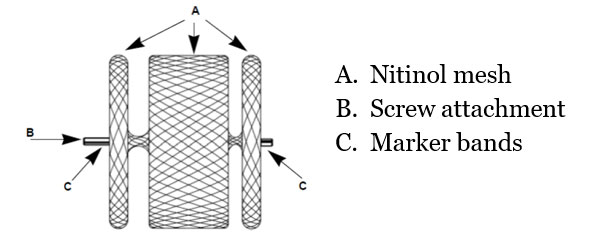
- Self-expanding device made from a Nitinol wire mesh
- The device is shipped attached to a 135cm nitinol delivery wire in a hoop dispenser
- The device is preloaded in a loader
- A plastic vise is also included and may be attached to the delivery wire to facilitate device detachment
Materials3:
- Plug: Nitinol
- Delivery wire: Nitinol
- Marker bands: Platinum
- Micro screw: Stainless steel
References
- Pech M, Kraetsch A, Winers G, et al., Embolization of the Gastroduodenal Artery Before Selective Internal Radiotherapy: A Prospectively Randomized Trial Comparing Platinum-Fibered Microcoils with the Amplatzer Vascular Plug II. CVIR 2009(32)3:455-61.
- AVP II is available in 8 unconstrained lengths ranging from 6 -18 mm.
- Amplatzer™ Vascular Plug II - Instructions for Use (IFU). Refer to IFU for additional information.
MAT-2200819 v2.0


