Advancing the standard of care in peripheral embolization
- A single device solution that allows for faster procedure times, less radiation exposure and lower procedural costs1,2
- Rapid embolization even in high-flow vessels1,3,5
- Designed for precise placement, controlled delivery and fully recapturable
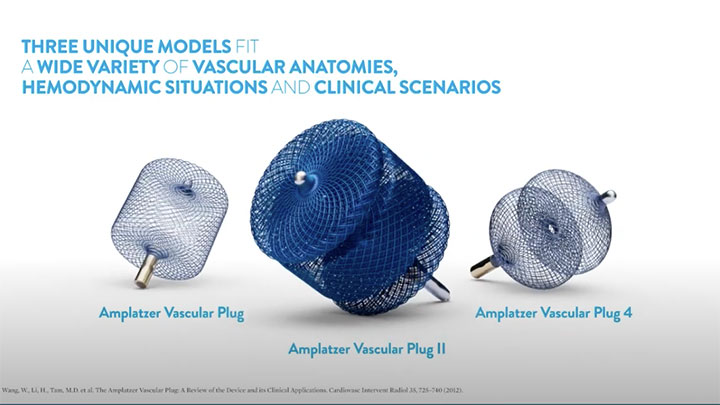
- A family of plugs for different vessel conditions
- Limited imaging artifact4
Amplatzer™ Vascular Plug (AVP)
- Single lobe, single layer device7
- Platinum marker bands on both ends7
- Select a device that is ~30-50% larger in diameter than the target vessel diameter7
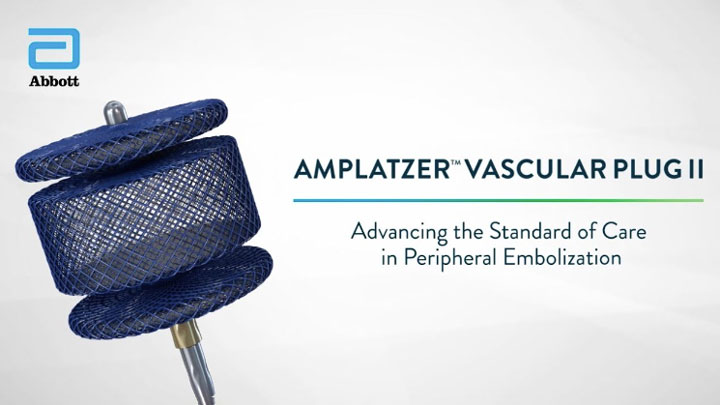
Amplatzer™ Vascular Plug II (AVP II)
- Multi-layered mesh designed to increase density and flow disturbance8
- Six planes of cross sectional coverage8 resulting in rapid occlusion1
- Select a device that is ~30-50% larger in diameter than the target vessel diameter8
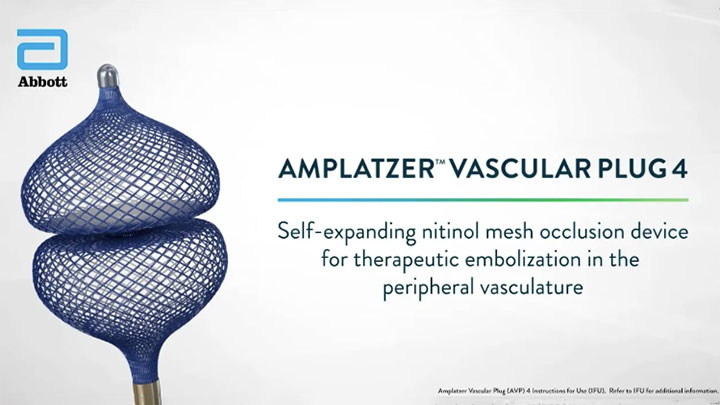
Amplatzer™ Vascular Plug 4 (AVP 4)
- Simple delivery through an 0.038” guide wire compatible diagnostic catheter9
- Easy tracking through tortuous anatomy6
- Extended reach to distal vasculature6
- Select a device that is ~30-50% larger in diameter than the target vessel diameter9
Amplatzer™ Vascular Plugs (AVP) Mechanism of Action
The nitinol mesh of an AVP acts as an embolic agent that promotes rapid clot formation. Each AVP creates a physical barrier to blood flow by instigating both the body’s immune response followed by a natural coagulation process.6
Indication / Intended Use
The Amplatzer™ Vascular Plug, Amplatzer™ Vascular Plug II, and Amplatzer™ Vascular Plug 4 are indicated for arterial and venous embolizations in the peripheral vasculature.
References
- Pech M, Kraetsch A, Wieners G, et al. Embolization of the Gastroduodenal Artery Before Selective Internal Radiotherapy: A prospectively Randomized Trial Comparing Platinum-Fibered Microcoils with the AMPLATZER Vascular Plug II. CVIR 2009(32)3:455-61.
- Hart JL, Aldin Z, et al. Embolization of pulmonary arteriovenous malformations using the AMPLATZER vascular plug: successful treatment of 69 consecutive patients. Eur Radiol 2010;20(11)2663-70.
- Kucukay F, Özdemir M, Şenol E, Okten S, Ereren M, Karan A. Large pulmonary arteriovenous malformations: long-term results of embolization with AMPLATZER vascular plugs. J Vasc Interv Radiol. 2014 Sep;25(9):1327-32. doi: 10.1016/j.jvir.2014.01.031. Epub 2014 Mar 18.
- Vandy F, Criado E, Upchurch GR, et al. Translumihnal Hypogastric Artery Occlusion with an Amplatzer Vascular Plug During Endovascular Aortic Aneurysm Repair. J Vasc Surg. 2008; 48(5)1121-4.
- Data on file at Abbott.
- Lopera, Jorge E. “The Amplatzer vascular plug: review of evolution and current applications.” Seminars in interventional radiology. Vol. 32. No. 04. Thieme Medical Publishers, 2015.
- Amplatzer™ Vascular Plug and Vascular Plug II Instructions for Use (IFU). Refer to IFU for additional information.
- Amplatzer™ Vascular Plug 4 Instructions for Use (IFU). Refer to IFU for additional information.
MAT-2405122 v1.0
Important Safety Information
AMPLATZER™ Vascular Plug

Intended Use: The AMPLATZER™ Vascular Plug is indicated for arterial and venous embolizations in the peripheral vasculature.
Contraindications: None known.
Warnings: The safety and effectiveness of this device for cardiac uses (for example, patent ductus arteriosus or paravalvular leak closures) and neurological uses have not been established.
Precautions:
Handling
- Patients allergic to nickel may suffer an allergic reaction to this device.
- This device was sterilized with ethylene oxide and is for single use only. Do not reuse or resterilize this device. Attempts to resterilize this device can cause a malfunction, insufficient sterilization, or harm to the patient.
- Use on or before the last day of the expiration month that is printed on the product packaging label.
- Do not use this device if the sterile package is open or damaged.
- This device should be used only by physicians who are trained in standard endovascular techniques. The physician should determine which patients are candidates for procedures that use this device.
- Store in a dry place.
MRI Safety Information:
| Device Name | Amplatzer™ Vascular Plug |
| Static Magnetic Field Strength (B0) | 1.5 T or 3.0 T |
| Maximum Field Spatial Gradient | 19T/m (1900 gauss/cm) |
| RF Excitation | Circularly Polarized (CP) |
| RF Transmit Coil Type | Body Coil |
| Operating Mode | Normal Operating Mode |
| Maximum Whole Body SAR | 2.0W/kg (Normal Operating Mode) |
| Maximum Head SAR | N/A |
| Scan Duration | 2.0W/kg whole-body-averaged SAR for 15 minutes of continuous scanning |
| MR Image Artifact | The presence of this implant may produce an image artifact. |
Potential Adverse Events: Potential complications include, but are not limited to: death, embolization of the device, hematoma at the site of entry, stroke, or vessel perforation, or embolization of the device.
State of California (USA) Only:
WARNING: This product can expose you to chemicals including ethylene oxide, which is known to the State of California to cause cancer and birth defects or other reproductive harm. For more information, go to www.P65Warnings.ca.gov.
MAT-2004315 v4.0
AMPLATZER™ Vascular Plug II

Intended Use: The AMPLATZER™ Vascular Plug II is indicated for arterial and venous embolizations in the peripheral vasculature.
Contraindications: None known.
Warnings: The safety and effectiveness of this device for cardiac uses (for example, patent ductus arteriosus or paravalvular leak closures) and neurological uses have not been established.
Precautions:
- This device was sterilized with ethylene oxide and is for single use only. Do not reuse or resterilize this device. Attempts to resterilize this device can cause a malfunction, insufficient sterilization, or harm to the patient.
- Use on or before the last day of the expiration month that is printed on the product packaging label.
- Patients with nickel allergy can experience an allergic reaction to this device.
- Do not use this device if the sterile package is open or damaged.
- This device should be used only by physicians who are trained in standard endovascular techniques. The physician should determine which patients are candidates for procedures that use this device.
- The physician should exercise clinical judgment in situations that involve the use of anticoagulants or antiplatelet drugs before, during, and/or after the use of this device.
- Store in a dry place.
MRI Safety Information:
| Device Name | Amplatzer™ Vascular Plug II |
| Static Magnetic Field Strength (B0) | 1.5 T or 3.0 T |
| Maximum Field Spatial Gradient | 19T/m (1900 gauss/cm) |
| RF Excitation | Circularly Polarized (CP) |
| RF Transmit Coil Type | Body Coil |
| Operating Mode | Normal Operating Mode |
| Maximum Whole Body SAR | 2.0W/kg (Normal Operating Mode) |
| Maximum Head SAR | N/A |
| Scan Duration | 2.0W/kg whole-body-averaged SAR for 15 minutes of continuous scanning |
| MR Image Artifact | The presence of this implant may produce an image artifact. |
Potential Adverse Events: Potential complications include, but are not limited to: death, migration of the device, stroke, or vessel perforation.
State of California (USA) Only:
WARNING: This product can expose you to chemicals including ethylene oxide, which is known to the State of California to cause cancer and birth defects or other reproductive harm. For more information, go to www.P65Warnings.ca.gov.
MAT-2004317 v4.0
AMPLATZER™ Vascular Plug 4

Indications And Usage: The AMPLATZER™ Vascular Plug 4 is indicated for arterial and venous embolizations in the peripheral vasculature.
Contraindications: None known.
Warnings:
- The safety and effectiveness of this device for cardiac uses (eg, cardiac septal occlusion, patent ductus arteriosus, paravalvular leak closures) and neurologic uses have not been established.
- Do not use this device if the sterile package is open or damaged.
- Use on or before the last day of the expiration month that is printed on the product packaging label.
- The device was sterilized with ethylene oxide and is for single use only. Do not reuse or resterilize this device. Attempts to resterilize this device can cause a malfunction, insufficient sterilization, or harm to the patient.
- Do not use a power injection syringe to inject contrast solution through this device.
- Physicians must be prepared to deal with urgent situations, such as device embolization, which require removal of the device. This includes the availability of an on-site surgeon.
Precautions:
- The AMPLATZER™ Vascular Plug 4 device consists of a nickel-titanium alloy, which is generally considered safe. However, in vitro testing has demonstrated that nickel is released from this device for a minimum of 60 days. Patients who are allergic to nickel may have an allergic reaction to this device, especially those with a history of metal allergies. Certain allergic reactions can be serious; patients should be instructed to notify their physicians immediately if they suspect they are experiencing an allergic reaction such as difficulty breathing or inflammation of the face or throat. Some patients may also develop an allergy to nickel if this device is implanted.
- The physician should exercise clinical judgment in situations that involve the use of anticoagulants or antiplatelet drugs before, during, and/or after use of the device.
- Store in a dry place.
- This device should be used only by physicians who are trained in standard endovascular techniques. The physician should determine which patients are candidates for procedures that use this device.
- Use in specific populations
- Pregnancy - care should be taken to minimize the radiation exposure to the
fetus and the mother. - Nursing mothers - there has been no quantitative assessment of the presence
of leachables in breast milk.
- Pregnancy - care should be taken to minimize the radiation exposure to the
MRI Safety Information:
| Device Name | Amplatzer™ Vascular Plug 4 |
| Static Magnetic Field Strength (B0) | 1.5 T or 3.0 T |
| Maximum Field Spatial Gradient | 19T/m (1900 gauss/cm) |
| RF Excitation | Circularly Polarized (CP) |
| RF Transmit Coil Type | Body Coil |
| Operating Mode | Normal Operating Mode |
| Maximum Whole Body SAR | 2.0W/kg (Normal Operating Mode) |
| Maximum Head SAR | N/A |
| Scan Duration | 2.0W/kg whole-body-averaged SAR for 15 minutes of continuous scanning |
| MR Image Artifact | The presence of this implant may produce an image artifact. |
Potential Adverse Events: Potential adverse events that may occur during or after a procedure placing this device include, but are not limited to:
- Air embolus
- Allergic reaction/toxic effects
- Bleeding
- Death
- Device migration
- Fever
- Foreign material embolic event
- Hemolysis
- Infection
- Occlusion of unintended vessel
- Peripheral embolism
- Recanalization
- Residual flow
- Stroke/TIA
- Surgical intervention
- Vascular access site complication
- Vessel trauma/perforation
State of California (USA) Only:
WARNING: This product can expose you to chemicals including ethylene oxide, which is known to the State of California to cause cancer and birth defects or other reproductive harm. For more information, go to www.P65Warnings.ca.gov.
MAT-2004318 v4.0