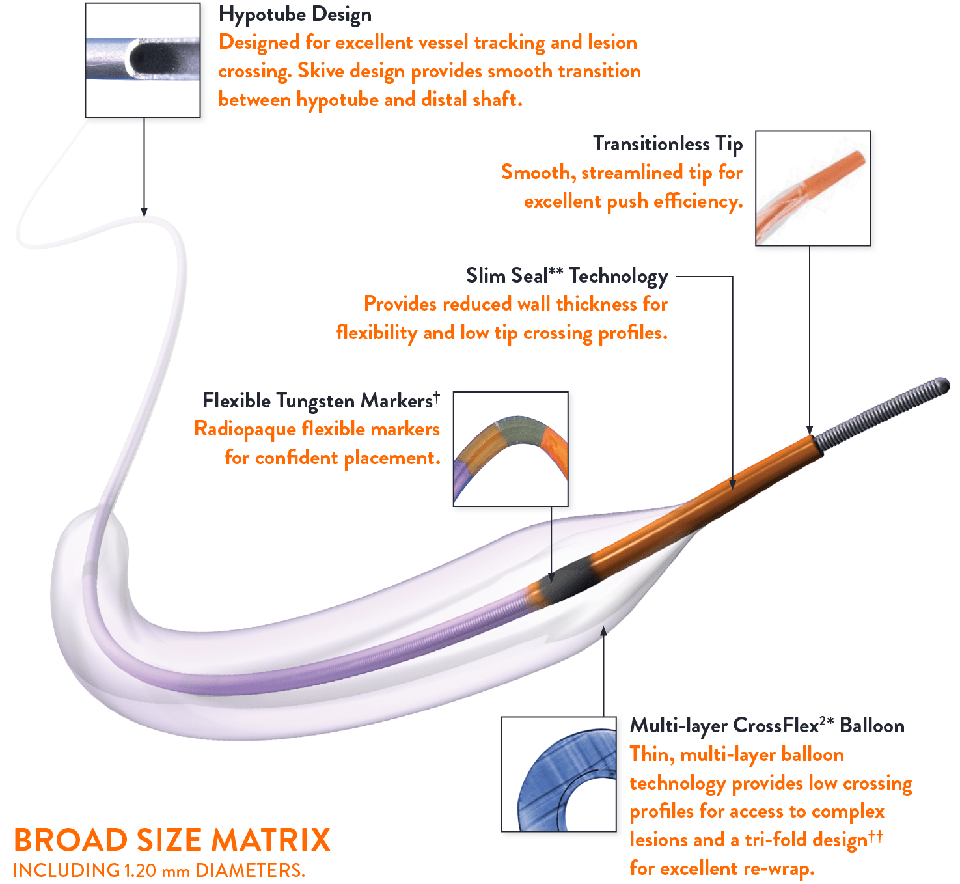
Transitionless Design for Challenging Anatomy
TREK™ Coronary Dilatation Catheter’s unique design provides smooth transitions from hub to tip.
- Transitionless tip
- Flexible distal shaft
- Multi-layer CrossFlex²* balloon and Slim Seal** technology
Mini Trek™ Coronary Dilatation Catheter Indicated for Treatment of De Novo Chronic Total Occlusions
MINI TREK™ Coronary Dilatation Catheter’s ultra low profile design enables lesion access.
- Small chassis designed distinctively for MINI TREK™ Coronary Dilatation Catheter
- Available in sizes as small as 1.20 mm
Technical Information
| Trek™ & Mini Trek™ Coronary Dilatation Catheters | |
|---|---|
| Platform | Rapid Exchange (RX) |
| Compliance | Semi-compliant |
| Nominal Pressure (NP) | 8 atm |
| Rated Burst Pressure (RBP) | 14 atm |
| Tip Entry Profile | 0.017 in (3.00 mm Ø) |
| Tip Crossing Profile | 0.021 in average (3.00 mm Ø) 0.023 in maximum |
| Folded Balloon Crossing Profile | 0.032 in average (3.00 mm Ø) 0.036 in maximum (3.00 mm Ø) |
| Refolded Balloon Crossing Profile | 0.056 in maximum (3.00 mm Ø) |
| RX Notch Diameter | 2.5 F average (3.00 mm Ø) 2.7 F maximum (all Ø) |
| Tip Length | <3 mm |
| Balloon Material | Pebax‡ |
| Marker Material | Flexible Tungsten/Pebax‡ |
| Catheter Length | 145 cm |
| Balloon Coating | Hydrophilic |
| Minimum Guide Catheter | 5 F (1.20 mm – 4.00 mm Ø) 6 F (4.50 mm & 5.00 mm Ø) |
| Maximum Guide Wire Compatibility | 0.014 in |
| Not Made With Natural Rubber Latex | Yes |
Ordering Information
Broad Size Matrix with 77 Sizes
| Balloon Diameter (mm) | Balloon Length (mm) | |||||||
|---|---|---|---|---|---|---|---|---|
| 6 | 8 | 12 | 15 | 20 | 25 | 30 | ||
| MINI TREK™ | 1.20 | 1012268-06 | 1012268-08 | 1012268-12 | 1012268-15 | 1012268-20 | - | - |
| 1.50 | 1012269-06 | 1012269-08 | 1012269-12 | 1012269-15 | 1012269-20 | - | - | |
| 2.00 | 1012270-06 | 1012270-08 | 1012270-12 | 1012270-15 | 1012270-20 | 1012270-25 | 1012270-30 | |
| TREK™ | 2.25 | 1012271-06 | 1012271-08 | 1012271-12 | 1012271-15 | 1012271-20 | 1012271-25 | 1012271-30 |
| 2.50 | 1012272-06 | 1012272-08 | 1012272-12 | 1012272-15 | 1012272-20 | 1012272-25 | 1012272-30 | |
| 2.75 | 1012273-06 | 1012273-08 | 1012273-12 | 1012273-15 | 1012273-20 | 1012273-25 | 1012273-30 | |
| 3.00 | 1012274-06 | 1012274-08 | 1012274-12 | 1012274-15 | 1012274-20 | 1012274-25 | 1012274-30 | |
| 3.25 | 1012275-06 | 1012275-08 | 1012275-12 | 1012275-15 | 1012275-20 | 1012275-25 | 1012275-30 | |
| 3.50 | 1012276-06 | 1012276-08 | 1012276-12 | 1012276-15 | 1012276-20 | 1012276-25 | 1012276-30 | |
| 3.75 | 1012277-06 | 1012277-08 | 1012277-12 | 1012277-15 | 1012277-20 | 1012277-25 | 1012277-30 | |
| 4.00 | 1012278-06 | 1012278-08 | 1012278-12 | 1012278-15 | 1012278-20 | 1012278-25 | 1012278-30 | |
| 4.50 | - | - | 1012279-12 | 1012279-15 | - | - | - | |
| 5.00 | - | - | 1012280-12 | 1012280-15 | - | - | - | |
Compliance Chart
| Balloon Diameter (mm) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pressure (atm) | 1.20 | 1.50 | 2.00 | 2.25 | 2.50 | 2.75 | 3.00 | 3.25 | 3.50 | 3.75 | 4.00 | 4.50 | 5.00 |
| MINI TREK™ | TREK™ | ||||||||||||
| 2 | 1.10 | 1.34 | 1.69 | 1.87 | 2.15 | 2.16 | 2.49 | 2.91 | 2.93 | 3.06 | 3.24 | 3.69 | 4.10 |
| 3 | 1.12 | 1.36 | 1.80 | 1.95 | 2.20 | 2.35 | 2.56 | 2.98 | 3.04 | 3.22 | 3.46 | 3.91 | 4.28 |
| 4 | 1.13 | 1.38 | 1.85 | 2.00 | 2.27 | 2.41 | 2.66 | 3.05 | 3.11 | 3.31 | 3.56 | 4.04 | 4.43 |
| 5 | 1.14 | 1.40 | 1.90 | 2.06 | 2.34 | 2.49 | 2.73 | 3.12 | 3.20 | 3.41 | 3.67 | 4.17 | 4.59 |
| 6 | 1.17 | 1.43 | 1.94 | 2.12 | 2.40 | 2.56 | 2.81 | 3.19 | 3.28 | 3.50 | 3.78 | 4.29 | 4.73 |
| 7 | 1.18 | 1.45 | 1.98 | 2.17 | 2.45 | 2.63 | 2.87 | 3.25 | 3.38 | 3.59 | 3.89 | 4.40 | 4.86 |
| 8 NP | 1.20 | 1.47 | 2.01 | 2.21 | 2.51 | 2.70 | 2.93 | 3.31 | 3.45 | 3.67 | 3.99 | 4.49 | 4.98 |
| 9 | 1.21 | 1.49 | 2.03 | 2.25 | 2.55 | 2.75 | 2.97 | 3.36 | 3.51 | 3.74 | 4.06 | 4.56 | 5.05 |
| 10 | 1.23 | 1.51 | 2.07 | 2.28 | 2.58 | 2.80 | 3.01 | 3.39 | 3.56 | 3.79 | 4.11 | 4.62 | 5.13 |
| 11 | 1.24 | 1.52 | 2.09 | 2.31 | 2.60 | 2.83 | 3.04 | 3.42 | 3.60 | 3.84 | 4.18 | 4.68 | 5.20 |
| 12 | 1.25 | 1.54 | 2.12 | 2.33 | 2.62 | 2.86 | 3.07 | 3.45 | 3.64 | 3.87 | 4.23 | 4.73 | 5.25 |
| 13 | 1.26 | 1.55 | 2.14 | 2.35 | 2.64 | 2.88 | 3.09 | 3.48 | 3.67 | 3.91 | 4.28 | 4.80 | 5.31 |
| 14 RBP | 1.27 | 1.56 | 2.17 | 2.37 | 2.67 | 2.91 | 3.11 | 3.50 | 3.70 | 3.94 | 4.32 | 4.86 | 5.36 |
| 15 | 1.28 | 1.57 | 2.19 | 2.39 | 2.69 | 2.92 | 3.14 | 3.53 | 3.73 | 3.99 | 4.38 | 4.92 | 5.41 |
| 16 | 1.29 | 1.59 | 2.21 | 2.41 | 2.71 | 2.96 | 3.16 | 3.56 | 3.75 | 4.02 | 4.44 | 4.98 | 5.50 |
| 17 | 1.30 | 1.60 | 2.24 | 2.43 | 2.73 | 2.99 | 3.18 | 3.58 | 3.78 | 4.05 | 4.49 | 5.04 | 5.56 |
| Nominal Pressure (NP) | Rated Burst Pressure (RBP) |
Data on file at Abbott.
*CrossFlex2 technology available on 2.25 mm – 5.0 mm balloon sizes.
**Slim Seal technology available on 2.0 mm – 3.75 mm balloon sizes.
† Single marker on all 6 mm lengths and 1.20 – 1.50 mm diameters (all lengths). Dual markers on all other sizes.
††Tri-fold design for 2.00 mm – 4.00 mm diameters. Bi-fold design for 1.20 mm & 1.50 mm diameters.
MAT-2501201 v2.0
TREK™ RX & OTW and MINI TREK™ RX & MINI TREK™ II OTW
Coronary Dilatation Catheters

Caution
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE. OBSERVE ALL WARNINGS AND PRECAUTIONS NOTED THROUGHOUT THESE INSTRUCTIONS. FAILURE TO DO SO MAY RESULT IN COMPLICATIONS.
Indications
Applies to TREK™ RX & OTW 2.25 mm – 5.00 mm sizes only:
The TREK™ RX & OTW Coronary Dilatation Catheters are indicated for:
- Balloon dilatation of the stenotic portion of a coronary artery or bypass graft stenosis, for the purpose of improving myocardial perfusion
- Balloon dilatation of a coronary artery occlusion, for the purpose of restoring coronary flow in patients with ST-segment elevation myocardial infarction
- Balloon dilatation of a stent after implantation
Applies to MINI TREK™ RX and MINI TREK™ II OTW
1.50 mm – 2.00 mm sizes only:
The TREK™ RX & OTW Coronary Dilatation Catheters are indicated for:
- Balloon dilatation of the stenotic portion of a coronary artery or bypass graft stenosis, for the purpose of improving myocardial perfusion
- Balloon dilatation of a coronary artery occlusion, for the purpose of restoring coronary flow in patients with ST-segment elevation myocardial infarction
- Balloon dilatation of a stent after implantation (balloon model 2.0 mm only)
- Balloon dilatation of de novo chronic total coronary occlusions (CTO)
Applies to MINI TREK™ RX and MINI TREK™ II OTW 1.20 mm sizes only:
The MINI TREK™ RX and MINI TREK™ II OTW 1.20mm Coronary Dilatation Catheters are indicated for:
- Initial balloon dilatation of the stenotic portion of a coronary artery or bypass graft stenosis (≥ 70% stenosis).
- Balloon dilatation of de novo chronic total coronary occlusions (CTO)
Note (applies to 2.00 mm to 5.00 mm only): Post-deployment stent expansion testing was performed on the bench with the MULTI-LINK VISION™ and MULTI-LINK ULTRA™ stents. All stents should be deployed in accordance with the manufacturer’s indications and instructions for use.
Contraindications (applies to all sizes)
The TREK™ RX & OTW, MINI TREK™ RX and MINI TREK™ II OTW Coronary Dilatation Catheters are not intended to be used to treat patients with:
- An unprotected left main coronary artery
- A coronary artery spasm in the absence of a significant stenosis
Warnings (applies to all sizes)
This device is intended for one time use only. DO NOT resterilize and / or reuse it, as this can compromise device performance and increase the risk of cross contamination due to inappropriate reprocessing.
Percutaneous transluminal coronary angioplasty (PTCA) should only be performed at hospitals where emergency coronary artery bypass graft surgery can be quickly performed in the event of a potentially injurious or life-threatening complication.
PTCA in patients who are not acceptable candidates for coronary artery bypass graft surgery requires careful consideration, including possible hemodynamic support during PTCA, as treatment of this patient population carries special risk.
Use only the recommended balloon inflation medium. Never use air or any gaseous medium to inflate the balloon.
Balloon pressure should not exceed the rated burst pressure (RBP). The RBP is based on results of in vitro testing. At least 99.9% of the balloons (with a 95% confidence) will not burst at or below their RBP. Use of a pressure-monitoring device is recommended to prevent overpressurization.
To reduce the potential for vessel damage, the inflated diameter of the balloon should approximate the diameter of the vessel just proximal and distal to the stenosis.
When the catheter is exposed to the vascular system, it should be manipulated while under high quality fluoroscopic observation. Do not advance or retract the catheter unless the balloon is fully deflated under vacuum. If resistance is met during manipulation, determine the cause of the resistance before proceeding.
Do not use, or attempt to straighten, a catheter if the shaft has become bent or kinked; this may result in the shaft breaking. Instead, prepare a new catheter.
Do not torque the catheter more than one (1) full turn.
Treatment of moderately or heavily calcified lesions is considered to be moderate risk, with an expected success rate of 60 – 85% and increases the risk of acute closure, vessel trauma, balloon burst, balloon entrapment, and associated complications. If resistance is felt, determine the cause before proceeding. Continuing to advance or retract the catheter while under resistance may result in damage to the vessels and / or damage / separation of the catheter.
In the event of catheter damage / separation, recovery of any portion should be performed based on physician determination of individual patient condition and appropriate retrieval protocol.
Precautions (applies to all sizes)
Note the “Use by” date specified on the package.
Inspect all product prior to use. Do not use if the package is open or damaged.
This device should be used only by physicians trained in angiography and PTCA, and / or percutaneous transluminal angioplasty (PTA).
Prior to angioplasty, the dilatation catheter should be examined to verify functionality and ensure that its size is suitable for the specific procedure for which it is to be used.
During the procedure, appropriate anticoagulant and coronary vasodilator therapy must be provided to the patient as needed. Anticoagulant therapy should be continued for a period of time to be determined by the physician after the procedure.
If the surface of the TREK™ RX & OTW, MINI TREK™ RX or MINI TREK™ II OTW Coronary Dilatation Catheter becomes dry, wetting with heparinized normal saline will reactivate the coating.
Do not reinsert the TREK™ RX & OTW, MINI TREK™ RX or MINI TREK™ II OTW Coronary Dilatation Catheter into the coil dispenser after procedural use.
The safety and effectiveness of this PTCA balloon catheter for the treatment of in-stent restenosis (ISR) have not been established.
Applies to TREK™ RX and MINI TREK™ RX only (APPLIES TO ALL SIZES), in addition to above:
The design and construction of these catheters do not provide the user with distal pressure monitoring capability.
Applies to TREK™ RX 4.50mm and5.00mm sizes only, in addition to above:
With 4.5 mm and 5.0 mm balloon dilatation catheters, some increased resistance may be noted upon insertion or withdrawal into or out of the guiding catheter. Choosing a larger guiding catheter size may minimize this.
Applies to TREK™ OTW and MINI TREK™ II OTW (APPLIES TO ALL SIZES), in addition to above:
Bench testing was conducted with 0.014” (.36mm) constant diameter guide wires to establish guide wire compatibility. If another type of guide wire is selected with a different dimensional profile, the compatibility (e.g., wire resistance) should be considered prior to use.
Adverse Events (applies to all sizes)
Possible adverse effects include, but are not limited to, the following:
- Acute myocardial infarction
- Arrhythmias, including ventricular fibrillation
- Arteriovenous fistula
- Coronary artery spasm
- Coronary vessel dissection, perforation, rupture, or injury
- Death
- Drug reactions, allergic reaction to contrast medium
- Embolism
- Hemorrhage or hematoma
- Hypo / hypertension
- Infection
- Restenosis of the dilated vessel
- Total occlusion of the coronary artery or bypass graft
- Unstable angina
MAT-2109405 v2.0
