Assert-IQ™ 3 Insertable Cardiac Monitor (ICM) is designed for dynamic office and clinic settings while encapsulating the essential and most valued ICM features. An innovative ICM – also commonly referred to as a loop recorder – the Assert-IQ 3 ICM offers:
- Advanced algorithms that offer enhanced accuracy in arrhythmia detection and time-to-diagnosis.1-3
- Clear, crisp EGMs to ensure superior P-wave visualization.4
- Leading Bluetooth® technology and SyncUP™ Remote Monitoring Support for timely and consistent diagnostic data.
Advanced Algorithms
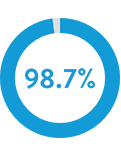
Reduce false detections by 98.7% for AF and Pause, while maintaining high sensitivity of
true EGMs.1, 2, 5
5-Step AF Detection Discriminator
Focuses on R-R interval patterns and P-waves in EGMs to determine if an event is true or false. Customize AF detection to patient-specific needs with programmable AF sensitivity.

Performance by the Numbers
Assert-IQ 3 ICM's AF algorithm reduces data burden by 21% compared to the latest version of LINQ II‡ while maintaining sensitivity.*, 1, 6, 7

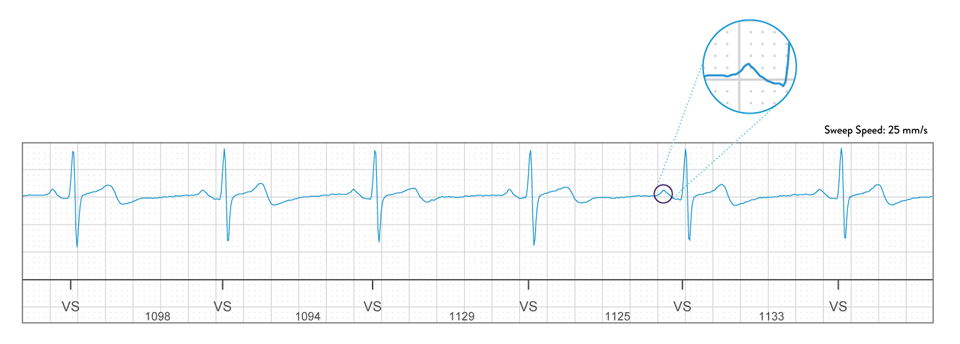
Clear, Crisp EGMs
Experience clear signal quality, making it easier for you to visualize P-waves and support quicker, more informed clinical decision making.4
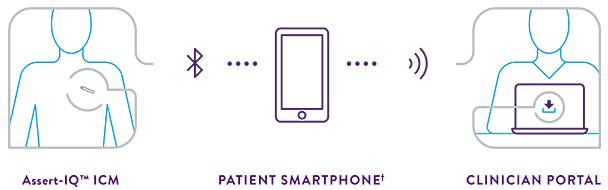
A Leader in ICM Bluetooth® Technology
Helping Patients Get Connected and Stay Connected
Leading ICM Bluetooth® technology helps your patients stay connected with ease on a daily basis. The myMerlin™ Mobile App automatically checks in with a connected device every 20 seconds to capture, encrypt and transfer data to the Merlin.net™ Patient Care Network (PCN).
Reliable Support for Remote Monitoring
SyncUP™ Remote Monitoring Support experts get your patients started with remote monitoring while reducing the time they need to stay post-procedure. Count on the remote connectivity team to provide free one-on-one education about remote monitoring to patients at home, pair their Assert-IQ 3 ICM to a smartphone, and streamline your outpatient implant workflow. Enrolled clinics receive a weekly report on remote patient monitoring compliance and increased accessibility to their Abbott representative for essential clinic needs.
Upon in-person visits, patients come in paired and knowledgeable about ICM remote monitoring.
Ordering Information
Assert-IQ 3 ICM
| MODEL# | DESCRIPTION | DIMENSIONS (H x L x T, MM) | LONGEVITY |
|---|---|---|---|
| DM5000 | Assert-IQ 3 ICM | 46.5 x 9.4 x 3.1 | 3 years |
Stay Informed
Sign up to hear about our technology, education opportunities, and more.
Read the Latest Blog Article
Stay up to date with recent news, product highlights, and case studies.
References
*As of 12.31.22 LINQ II‡ with AccuRhythm‡ AI. EGM burden comparison is based on two independent, random, real-world data sets. Patient characteristics and device programming may differ.
- Gopinathannair R, Lakkireddy D, Manyam H, et al. Improving the Specificity of Atrial Fibrillation and Tachycardia Detection in an Insertable Cardiac Monitor. Presented at Heart Rhythm Society (HRS); San Francisco, USA; 2022.
- Afzal MR, Gopinathannair R, Manyam H, et al. Development and Evaluation of a New Algorithm Enhancement to Improve Specificity of Pause Detection in an Insertable Cardiac Monitor. Presented at Heart Rhythm Society (HRS); San Francisco, USA; 2022.
- Gardner RS, Quartieri F, Betts TR, et al. Reducing the Electrogram Review Burden Imposed by Insertable Cardiac Monitors. J Cardiovascular Electrophysiology. 2022;33(4):741-750. doi:10.1111/jce.15397
- Shehata MM, Nair DG, Qu F, et al. Insertable Cardiac Monitor P-wave Visibility in a New Clinical Report. Presented at Asia Pacific Heart Rhythm Society (APHRS); Bangkok Thailand; 2022.
- Data on File. Abbott - Report SJM-CFM-0919-0163.
- Radtke, et al. Artificial Intelligence Enables Dramatic Reduction of False Atrial Fibrillation Alerts from Insertable Cardiac Monitors. Presented at Heart Rhythm Society (HRS); Boston, USA; Aug 2021.
- Data on File. Abbott - Report 90986479A; AF EGM Burden Reduction in Assert-IQ ICM.
™ Indicates a trademark of the Abbott group of companies.
‡ Indicates a third-party trademark, which is property of its respective owner.
Bluetooth and Bluetooth logo are registered trademarks of Bluetooth SIG, Inc.
MAT-2314413 v2.0

