The Assert-IQ™ Insertable Cardiac Monitor (ICM) is now the only ICM system to combine AI, the longest-lasting Bluetooth® ICM* and IQ Insights to optimize patient management across arrhythmia types.1-11
It features:
- Advanced AI algorithms that reduce false positives from AF and Pause episodes while maintaining high sensitivity.4
- Extended monitoring with the longest-lasting Bluetooth® ICM‡ with full functionality and no compromises.
- Clear, crisp EGMs for improved visualization of P-waves.3
- 1.5 Tesla (T) and 3T MR Conditional.
The Assert-IQ ICM system leverages AI to enhance the accuracy of AF and Pause episode detection, significantly reducing data burden for clinicians.4
Longest-Lasting Bluetooth® ICM†
Assert-IQ ICM is a Bluetooth®-enabled ICM that provides reliable, long-lasting cardiac monitoring with no compromises in features.
Assert-IQ™ EL+
6+ years** may be preferred for longer term monitoring, such as those undergoing therapy and at risk of developing further arrhythmias such as AF.
Assert-IQ™ 3/3+
3 years may be preferred for more traditional reasons for monitoring, such as diagnosing syncope, palpitations, or detection of atrial fibrillation (AF) within cryptogenic stroke patients.



New IQ Insights
Intelligent diagnostics provide trends and timelines of clinically relevant information to provide a holistic view for patient management and empower data-driven decisions.
Premature
Ventricular
Contractions
(PVCs)
Elevated Heart Rate with and without Activity
Body Position &
Posture Changes
DirectTrend™ Viewer



Remote Programmability
Assert-IQ ICM offers simple and secure remote programming, allowing you to control any connected device without requiring your patients to physically visit the clinic.*** This can reduce clinic workflow burden and ensure transmitted data is unique to the patient.
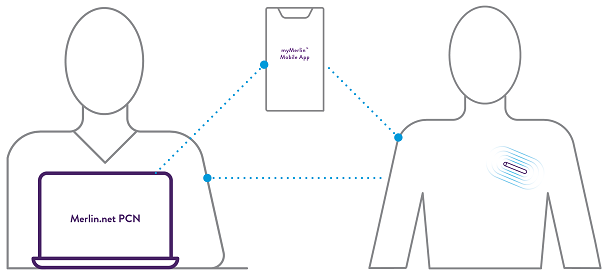
All parameters can be remotely programmed by the clinician.^
Assert-IQ ICM: The Latest Cardiac Monitoring Innovation
References
†As of 12.31.23. Reveal LINQ‡ User Manual, LINQ II
‡ User Manual, Lux Dx‡ User Manual, Biomonitor III‡ User Manual, Biomonitor IIIm‡ User Manual, Biomonitor IV‡ User Manual.
*DM5500 Model.
**Data on File, Abbott - Report 90984075.
***Available on DM5300/DM5500.
^Not applicable for Name, DOB, and reason for monitoring
- Gopinathannair R, Shehata MM, Afzal MR, et al. Novel Algorithms Improve Arrhythmia Detection Accuracy in Insertable Cardiac Monitors. Journal of Cardiovascular Electrophysiology. 2023;34(9):1961-1968. doi:10.1111/jce.16007
- Gardner RS, Quartieri F, Betts TR, et al. Reducing the Electrogram Review Burden Imposed by Insertable Cardiac Monitors. J Cardiovascular Electrophysiology. 2022;33(4):741-750. doi:10.1111/jce.15397
- Shehata MM, Nair DG, Qu F, et al. Insertable Cardiac Monitor P-wave Visibility in a New Clinical Report. Presented at Asia Pacific Heart Rhythm Society (APHRS); Bangkok Thailand; 2022.
- Abbott. Assert-IQ™ ICM and Merlin.net Artificial Intelligence for use with Assert-IQ™ ICM User Manuals.
- Medtronic. REVEAL LINQ‡ LNQ11 Insertable Cardiac Monitor and Patient Assistant PA96000 Clinician Manual. Updated January 25, 2022. Accessed February 23, 2024. https://www.medtronic.com/content/dam/emanuals/crdm/M027641C001B_view.pdf
- Medtronic. LINQ II‡ LNQ22 Insertable Cardiac Monitor Clinician Manual. Updated October 11, 2022. Accessed February 23, 2024. https://www.medtronic.com/content/dam/emanuals/crdm/M027638C001B_view.pdf
- Boston Scientific. User’s Manual, LUX-Dx‡ Insertable Cardiac Monitor System M301, 2925, 2935. Updated May 2022. Accessed February 23, 2024.
- Boston Scientific. User’s Manual, LUX-Dx II/II+‡ Insertable Cardiac Monitor System M302, M312, 2925, 2929, 2935, 2939. Updated August 2023. Accessed February 23, 2024.
- Biotronik. Technical Manual BIOMONITOR III‡. Updated March 22, 2023. Accessed February 23, 2024. https://manuals.biotronik.com/emanuals-professionals/?country=ES&product=ImplCardMon/BioMonitor3/BioMonitor3
- Biotronik. Technical Manual BIOMONITOR IIIm‡. Updated March 22, 2023. Accessed February 23, 2024. https://manuals.biotronik.com/emanuals-professionals/?country=ES&product=ImplCardMon/BioMonitor3m/BioMonitor3m
- Biotronik. Technical Manual BIOMONITOR IV‡. Updated December 06, 2023. Accessed February 23, 2024. https://manuals.biotronik.com/emanuals-professionals/?country=ES&product=ImplCardMon/BioMonitor4/BioMonitor4
Additional references
- Medtronic. LINQ II‡ LNQ22 Insertable Cardiac Monitor Clinician Manual. Updated September 01, 2022. Accessed February 23, 2024. https://www.medtronic.com/content/dam/emanuals/crdm/M032283C001B_view.pdf
- Boston Scientific. User’s Manual, LUX-Dx‡ Insertable Cardiac Monitor System M301, 2925, 2935. Updated April 2023. Accessed February 23, 2024. https://www.bostonscientific.com/content/dam/elabeling/crm/51656369-001_LUX-Dx_ICM_UM_en_S.pdf
- Boston Scientific. User’s Manual, LUX-Dx II/II+‡ Insertable Cardiac Monitor System M302, M312, 2925, 2929, 2935, 2939. Updated August 2023. Accessed February 23, 2024. https://www.bostonscientific.com/content/dam/elabeling/crm/51583079-001_LUX-Dx_ICM_UM_en_S.pdf
MAT-2403093 v5.0
