For a Full Physiology Diagnosis3-5
- Make a full physiology assessment with clearly visualized data needed to make decisions.1
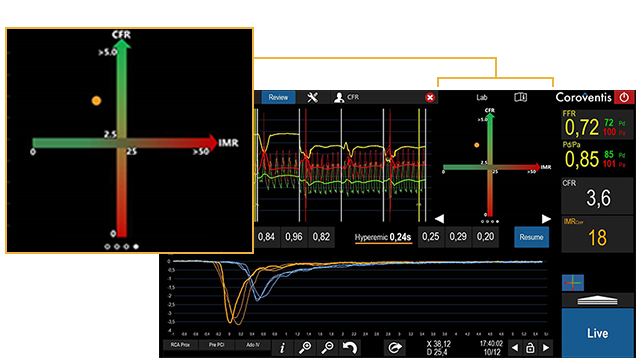
- Routine display of CFR/IMR values simplified for new users starting out CMD assessments
- Results Matrix to uncomplicate data interpretation and guide CMD decision making for new users
- Full physiology performance insights readily available with the Statistics Tab to inform cath lab utilization decisions and hospital marketing.1

Providing Hemodynamic Data for Entire Coronary Vasculature
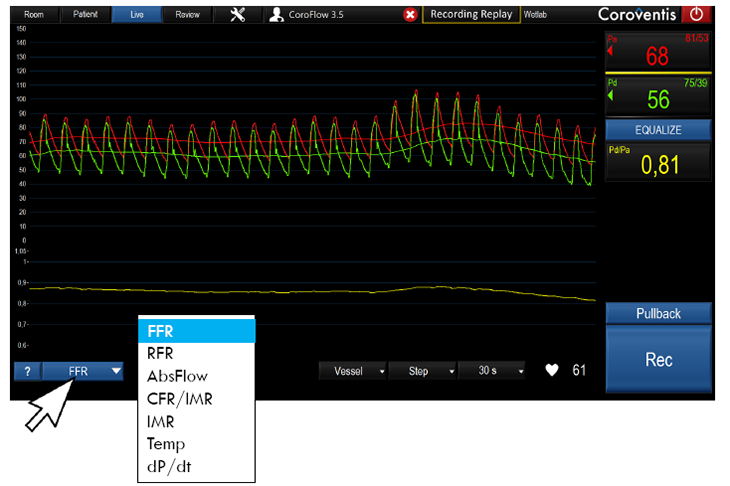
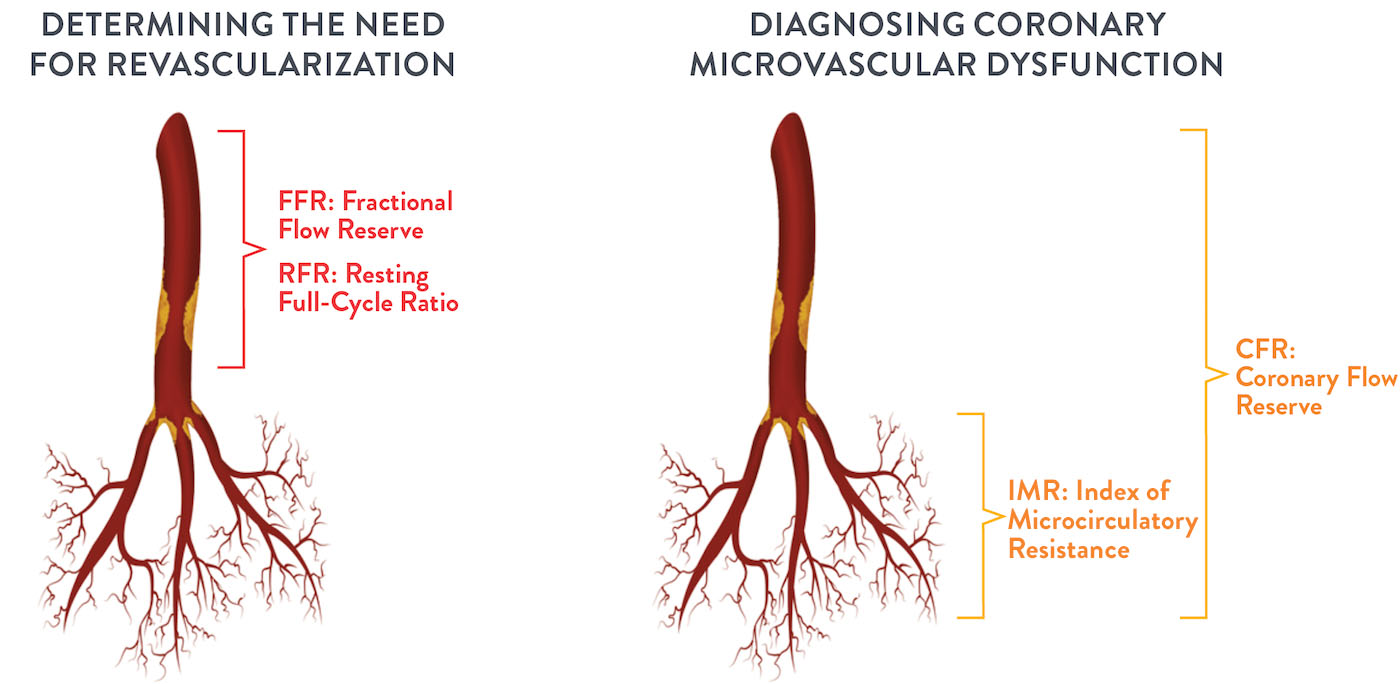
The CoroFlow‡ Cardiovascular System is an advanced platform to measure physiological indices: fractional flow reserve (FFR) and resting full-cycle ratio (RFR) to assess epicardial vessels; plus coronary flow reserve (CFR) and index of microcirculatory resistance (IMR) to assess microcirculation. This system is designed specifically to communicate with the wireless PressureWire™ X Guidewire.1,2
The CoroFlow‡ Cardiovascular System is capable of calculating and displaying hemodynamic measurements in both epicardial vessels and coronary microvasculature. Moreover, 50-65% of patients with angina with non-obstructive coronary artery disease (NOCAD) are believed to have Coronary Microvascular Dysfunction (CMD).6 Give patients a clear diagnosis of CMD and take the first step to improve their quality of life.4
CoroFlow‡ Cardiovascular System provides physiology data for vessel characteristics throughout the entire coronary vasculature.
Questions?
We are proud of the quality of our products and services, and remain committed to providing exceptional support and assistance to our customers across the globe.
For ordering information, visit ordering.
For technical issues, answers to frequently asked questions of a technical nature, and troubleshooting options, visit support.
References
- CoroFlow‡ Cardiovascular System Instructions for Use (IFU). Refer to IFU for additional information.
- PressureWire™ X Guidewire Instructions for Use (IFU). Refer to IFU for additional information.
- Ford TJ, Stanley B, Sidik N, et al. 1-year outcomes of angina management guided by invasive coronary function testing (CorMicA). JACC Intv. 2020; 13:33-45.
- Pijls NH, Fearon WF, Tonino PA, et al. Fractional Flow Reserve Versus Angiography for Guiding Percutaneous Coronary Intervention in Patients with Multivessel Coronary Artery Disease. 2-Year Follow-Up of the FAME (Fractional Flow Reserve Versus Angiography for Multivessel Evaluation) Study. JACC. 2010; 56(3): 177-184.
- Data on file at Abbott
- Marinescu MA, Löffler AI, Ouellette M, et al. Coronary microvascular dysfunction and microvascular angina: a systematic review of therapies. JACC Cardiovasc Imaging. 2015;8:210-220. doi:10.1016/j.jcmg.2014.12.008.
MAT-2006711 v3.0
PressureWire™ X Guidewire

Indications: The PressureWire™ X Guidewire is indicated to direct a catheter through a blood vessel and to measure physiological parameters in the heart and in the coronary and peripheral blood vessels. Physiological parameters include blood pressure. The PressureWire™ X Guidewire can also measure blood temperature.
Contraindications: This guidewire is contraindicated for use in the cerebral vasculature.
Warnings:
- No modification of this device is allowed.
- The PressureWire™ X Guidewire is supplied sterile. Discard the guidewire if the pouch is opened or damaged, compromising the sterile barrier. The guidewire is designed for single use only and shall not be reused or resterilized. Adverse effects of using a non-sterile or resterilized guidewire may include, but are not limited to:
- Local and/or systemic infection
- Mechanical damage
- Inaccurate readings
- Observe all guidewire movements. Whenever the guidewire is moved or torqued, the tip movement should be examined under fluoroscopy. Never push, withdraw, or torque the guidewire if it meets resistance or without observing corresponding movement of the tip, otherwise vessel/ventricle trauma may occur.
- Torquing or excessive manipulation of the guidewire in a sharp bend, against resistance, or repeated attempts to cross a total vessel occlusion may:
- Cause dissection or perforation of blood vessels
- Cause vessel spasm
- Damage and/or fracture the guidewire
- When introducing the guidewire, flush the catheter and administer anticoagulation as for a standard catheterization procedure or clotting may occur.
- Do not use the guidewire in the ventricles if the patient has a prosthetic mechanical or biological valve. It may result in damage to both the prosthesis and the guidewire, which may cause injury or death.
- Use of the PressureWire™ X Guidewire in conjunction with interventional devices with a short rapid exchange may result in a folded or fractured guidewire.
- High frequency surgical devices must not be used on a patient at the same time as the guidewire.
Precautions:
- The PressureWire™ X Guidewire is a delicate instrument and should be handled carefully.
- Make sure that the transmitter is kept dry to ensure accurate pressure and/or temperature readings. Inaccurate readings may necessitate device replacement.
- Do not use the guidewire in conjunction with atherectomy catheters. It may damage the guidewire.
- Do not withdraw or manipulate the guidewire in a sharp-edged object. It may result in abrasion of the guidewire coating.
- Factors that may affect the accuracy of the diagnostic information include, but are not limited to:
- Improper placement of the aortic pressure sensor.
- Failure to achieve maximum coronary and myocardial hyperemia in FFR procedures.
- Blood flow affected by the position of interventional devices, such as balloon catheters.
- Guidewire readings may be affected by defibrillation. Rezero the guidewire after defibrillation use.
- Do not measure pressure when the guidewire sensor element is in a sharp bend or in contact with atrial or ventricular walls. It might result in pressure artifacts.
- Do not use the PressureWire™ X Guidewire together with another guidewire, for so called jailed wire technique, due to difficulty in guidewire withdrawal and possible guidewire entrapment.
- Store at room temperature (15°C – 25°C) in a dry and dark place.
Potential Adverse Events: Potential complications which may be encountered during all catheterization procedures include, but are not limited to: vessel dissection or occlusion, perforation, embolus, spasm, local and/or systemic infection, pneumothorax, congestive heart failure, myocardial infarction, hypotension, chest pain, renal insufficiency, serious arrhythmias, or death.
In addition, this device has a coating containing Polyethylene Glycol (PEG); potential allergic reactions (anaphylaxis) may occur during the interventional procedure if the patient is allergic to PEG.
MAT-2103599 v2.0

Stay Connected