Pioneering Innovation in LBBAP
Experience the innovation of the UltiPace Pacing Lead – Abbott's latest breakthrough and FDA-approved stylet-driven lead for left bundle branch area pacing (LBBAP). Engineered for superior performance in the pursuit of continuous innovation.1

Connect with your Abbott Representative to learn more about LBBAP and stay informed about training opportunities. You can also sign up to receive updates on UltiPace Pacing Lead LBBAP clinical advancements.
Proven Design Evolved
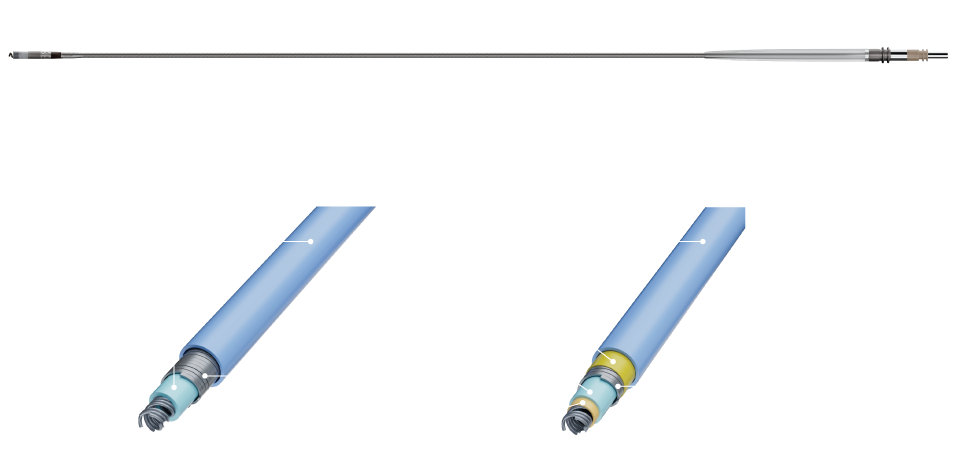
vs. Tendril™ STS1
3X
Increase in abrasion resistance
3.5X
Increase in lead crush resistance
vs. Tendril MRI™ 1
2.2X
Increase in abrasion resistance
2.6X
Increase in lead crush resistance
Demonstrated LBBAP Success and Safety
Composite
success
rate3*
of patients not experiencing
LBBAP-related adverse effects at 6 months post-implant.3
Resistant to Distal Fatigue Failure
UltiPace Pacing Leads have a robust distal lead tip without a flexible zone that helps reduce stress on the conductors and the chance for distal fatigue failure compared to competitive leads.4

Zero Helix Retraction
The Helix Locking Tool is a proven solution to lock an extended helix during tissue burrowing without retraction.4

The Helix Locking Tool aids with lead fixation by providing control over the extension and retraction of the helix.
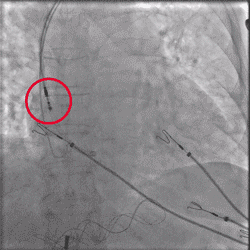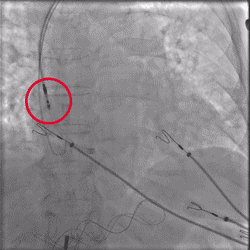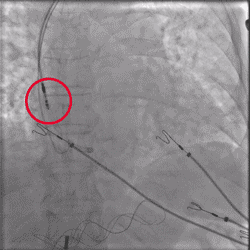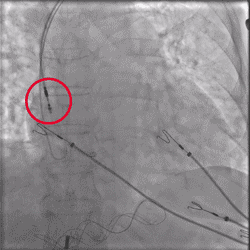
Improved Torque Transmission with Fewer Rotations
Improvement in torsional stiffness when using UltiPace Pacing Leads compared to LBBAP-approved lumenless pacing leads.4
Torque being applied to UltiPace lead body in LBBAP procedure.
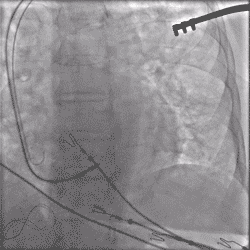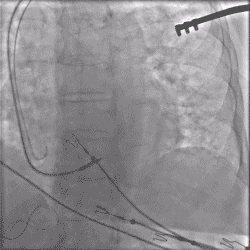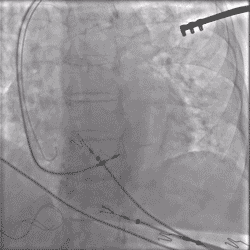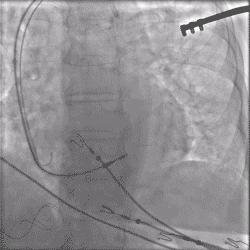
Continuous Unipolar Pacing and Impedance Monitoring
Provides the user with uninterrupted feedback to help achieve optimal results.5
Stay Informed
Sign up to hear about our technology, education opportunities, and more.
Read the Latest Blog Article
Stay up to date with recent news, product highlights, and case studies.
References
*Composite success rate of acceptable capture thresholds and sense amplitudes for LBBAP at 6 months post-implant.
- Abbott. Data on File. Item: 91010074 Ver A.
- Vijayaraman P, Sharma PS, Atwater BD, Nair D, Joza J, Zanon F, Li W, Kim JA, Nabutovsky Y, Lee K, Schaller RD. Two-year follow-up of the safety and clinical outcomes of stylet-driven leads for left bundle branch area pacing compared with right ventricular pacing in the Medicare population. Heart Rhythm. 2025 Jul 9:S1547-5271(25)02633-5.
- Abbott. Left Bundle Branch Area Pacing Study. Item: ARTEN600334504 A.
- Abbott. Data on File, Engineering Technical Report. Item: 90994484 Ver D.
- Burri, H., Jastrzebski, M., Cano, O., Curila, K., Pooter, J. de, Huang, W., Israel, C., Joza, J., Romero, J., Vernooy, K., Vijayaraman, P., Whinnett, Z., & Zanon, F. (2023). EHRA clinical consensus statement on conduction system pacing implantation: Executive summary. endorsed by the Asia-Pacific Heart Rhythm Society (APHRS), Canadian heart rhythm society (CHRS) and Latin-American heart rhythm society (LAHRS). Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. https:// pubmed.ncbi.nlm.nih.gov/37061850/
Indications:
UltiPace™ leads are indicated for use in combination with a compatible pacemakers, implantable cardioverter defibrillator (ICDs) or cardiac resynchronization therapy (CRT-P/CRT-D) to provide sensing and pacing for the management of chronic symptomatic bradycardia and various atrioventricular conduction abnormalities in patients who experience syncope, presyncope, fatigue, disorientation due to arrhythmia/ bradycardia, or any combination of these symptoms. The UltiPace leads are implanted transvenously in either the right atrium, the right ventricle or the left bundle branch area.
MAT-2403244 v5.0



