Evolve to Epic
For decades, Epic™ Tissue Valves have been the surgical valve platform that cardiac surgeons and patients have relied on for more flexibility and more possibilities.
Built on a proven legacy, with decades of durability data, Epic Valves deliver on everything you expect from a tissue heart valve. With key features like porcine leaflets, radiopaque markers, and fracturable stent—Epic Valves are prepared for a potential future intervention.
Join the Epic Evolution.
Learn about the key features of Epic™ tissue valves.
Ordering Information
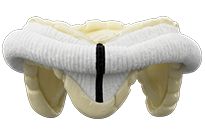
Epic™ Plus Mitral Valve
| Model Number | Valve Size (mm) | Tissue Annulus Diameter (mm) | Internal Diameter (Stent ID mm) | Ventricular Protrusion (mm) | Total Height (mm) | Cuff Outer Diameter (mm) |
|---|---|---|---|---|---|---|
| E200-25M | 25 | 25 | 22.6 | 9 | 16 | 33 |
| E200-27M | 27 | 27 | 24.5 | 9 | 17 | 35 |
| E200-29M | 29 | 29 | 26.3 | 10 | 19 | 37 |
| E200-31M | 31 | 31 | 28.4 | 10 | 20 | 39 |
| E200-33M | 33 | 33 | 30.3 | 11 | 20 | 41 |
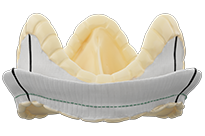
Epic™ Plus Supra Aortic Valve
| Model Number | Valve Size (mm) | Tissue Annulus Diameter (mm) | Internal Diameter (Stent ID mm) | Aortic Protrusion (mm) | Total Height (mm) | Cuff Outer Diameter (mm) |
|---|---|---|---|---|---|---|
| ESP200-19 | 19 | 19 | 18.7 | 11 | 14 | 25 |
| ESP200-21 | 21 | 21 | 20.8 | 11 | 15 | 28 |
| ESP200-23 | 23 | 23 | 22.6 | 13 | 16 | 29 |
| ESP200-25 | 25 | 25 | 24.5 | 13 | 17 | 31 |
| ESP200-27 | 27 | 27 | 26.3 | 14 | 19 | 33 |
Accessories
| Model Number | Description |
|---|---|
| E2000 | Bioprosthetic Heart Valve Sizer Set One (1) click-in Aortic holder handle One (1) Screw-in holder handle Six (6) double-ended aortic/Supra sizers (19, 21, 23, 25, 27, 29) Five (5) mitral sizers (25, 27, 29, 31, 33) One (1) autoclavable tray for storage of components One (1) autoclavable tray cover |
| E2000-HA | Replacement handle One (1) click-in Aortic holder handle |
| E2000 HM | Replacement handle One (1) Screw-in Mitral holder handle |
EPIC™ PLUS/ EPIC™ PLUS SUPRA STENTED PORCINE TISSUE VALVES

Indications for Use
The Epic™ Plus valve is indicated for patients requiring replacement of a diseased, damaged, or malfunctioning native aortic and/or mitral heart valve. It may also be used as a replacement for a previously implanted aortic and/or mitral prosthetic heart valve.
The Epic™ Plus Supra valve is indicated for patients requiring replacement of a diseased, damaged, or malfunctioning native aortic heart valve. It may also be used as a replacement for a previously implanted aortic prosthetic heart valve.
Contraindications
None known.
Potential Adverse Events
Adverse events potentially associated with the use of bioprosthetic heart valves (in alphabetical order) include: angina; cardiac arrhythmias; endocarditis; heart failure; hemolysis; hemolytic anemia; hemorrhage, anticoagulant/antiplatelet-related; leak, transvalvular or paravalvular; myocardial infarction; nonstructural dysfunction (entrapment by pannus or suture, inappropriate sizing or positioning, or other); prosthesis regurgitation; stroke; structural deterioration (calcification, leaflet tear, or other); thromboembolism; valve thrombosis.
It is possible that these complications could lead to: reoperation; explantation; permanent disability;
death.
MAT-2607066 v3.0
