Precise, Efficient Hemostasis.
Precise Placement and Control
- Integrated manometer allows pressure to be adjusted based on patient status.
- The inflatable, transparent dome offers precise pressure for effective hemostasis.
- Adjustable belt designed to fit securely so small patient movements may not cause the device to slip.

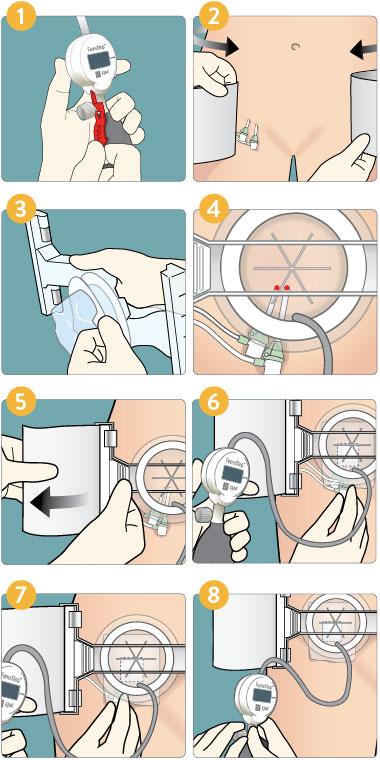
Deployment
- Pull the red tab to activate the pump.
- Position belt in line with puncture site(s), equal on both sides.
- Peel back lid, keeping the dome sterile.
- Center dome over arterial puncture (superior and medial to skin incision). Keep sheath hubs clear of dome rim.
- Attach belt to arch and fit snug. Ensure arch is level and square across the groin area. Adjust by pulling on appropriate corner of belt to make arch perpendicular to femoral compression site.
- Inflate dome to 20-30 mmHg; then remove venous sheath* (if applicable).
- Further inflate to 60-80 mmHg and remove arterial sheath (if applicable). Then quickly increase pressure in dome to be +10-20 mmHg greater than systolic BP or higher as necessary to maintain initial hemostasis.
- After 3 minutes maximum; lower to a maintenance pressure until limb profusion is restored to baseline and hemostasis is maintained. Check pedal pulse periodically to confirm whether or not flow remains in vessel. After appropriate duration as defined by hospital guidelines; lower by 10-20 mmHg every few minutes until you reach zero. Leave in place at low pressure if appropriate. Remove carefully. Dress wound. Discard device.
Efficient Workflow
- Hands-free femoral artery compression allows staff to monitor multiple patients at one time.
- Transparent dome ensures puncture site visibility.
- Hands-free compression minimizes staff exposure to blood and reduces neck, arm, and wrist fatigue.
Ordering Information
| FemoStop™ Gold Femoral Compression System | |||
|---|---|---|---|
| Description | Stock Number | Units Per Package | Includes |
| FemoStop™ Gold | C11165 | 1 | (1) Arch with Pneumatic Dome (1) Integrated Digital Manometer (1) Adjustable Belt (1) Pinch Clamp |
FemoStop™ Gold Femoral Compression System — Instructions For Use (IFU). Refer to the IFU for additional information.
MAT-2009620 v3.0
FemoStop™ Gold Femoral Compression System

Indications For Use
The FemoStop™ Femoral Compression System is indicated for use in the compression of the femoral artery or vein after vessel cannulation and in ultrasound-guided compression repair of a femoral artery pseudoaneurysm.
Contraindications
- Severe peripheral vascular disease due to the risk of arterial thrombosis.
- Critical limb ischemia.
- Overlying skin necrosis and/or infection.
- Arterial injuries above or near the inguinal ligament.
- The inability to adequately compress due to e.g. coexisting very large hematomas, excessive pain or discomfort (despite anesthetics/analgesics).
- Patients not suitable for compression of their femoral artery due to leg edema, femoral nerve compression, or arterial obstruction.
- Femoral artery graft or vein graft due to the risk of damage.
- Ultrasound-guided compression repair of infected femoral pseudoaneurysms.
Warnings
- For one time use only. Do not reuse or resterilize. Do not use if the original sterile package is not intact. Inspect the system carefully prior to use to verify that all parts are present and undamaged.
- Reuse after cleaning attempts, resterilization and repackaging may result in patient/user infections, product deterioration leading to, e.g. reduced concentration of dome pressure, causing bleeding. Do not disassemble or attempt to repair the system.
- Adequate compression may not be obtained in markedly obese patients.
- Avoid releasing pressure suddenly to reduce any risk of flushing thrombotic material into the artery. Release of thrombotic material may result in embolization which could lead to patient injury.
- Do not leave the system on the patient for inappropriately long compressions, as tissue damage may occur. A brief interruption at least every three hours of pressure is recommended during long compression periods. Inappropriately long compression and/or immobilization may increase the risk for thrombosis or embolization which could lead to patient injury or death.
- If arterial/venous hemostasis is not achieved, significant bleeding may occur which could result in patient injury or death.
- While removing the sheath, ensure that the pressure applied is kept low, to avoid damage to the vessel or a “milking” effect. Allowance for slight bleeding at the site is preferred to preclude introduction of thrombus to the vessel.
- To prevent limb ischemia, do not leave artery completely blocked for more than 3 minutes. Check pedal pulse periodically to confirm whether or not flow remains in the vessels.
- To minimize the risk for arterial/venous fistula formation, venous hemostasis should be achieved prior to removal of the arterial sheath.
- Do not apply pressure to a femoral artery stent due to risk of damage.
Precautions
- The FemoStop™ Femoral Compression System should only be used for compression of the femoral artery or vein after vessel cannulation by or on the order of physicians trained in femoral artery or vein compression procedures.
- Federal (USA) law restricts this device to sale by or on the order of a physician.
- When removing the sheath for an Intra-Aortic Balloon Pump (IABP), the Instruction for Use for the IABP should be followed as appropriate.
- For successful compression, the system must be snug and secure around the patient’s hips before pressure is applied. Do not over-tighten the belt.
- For successful compression, the system must be correctly positioned throughout the procedure so that pressure is applied to the point intended.
- On very obese patients, it may be necessary to tighten the belt slightly more to enhance downward compression.
- When using the system on obese patients, fatty tissue may be displaced giving a false impression of a developing hematoma.
- Placement of the system may not be suitable on large patients, or patients with very wide hips as the belt may be too short. An abdominal strap/tape may be used to pull excessive adipose tissue away from dome.
- The target inflation pressure should be 10-20mmHg above the systolic pressure, or higher if necessary to control the bleeding. Exceeding pressures of 200mmHg may indicate the need to tighten the belt or reposition the dome.
- Careful monitoring of the dome pressure during the initial period of use is recommended, as the elastic material of the dome may stretch slightly during the first few minutes. You may notice a slight drop in pressure on the manometer. If this occurs, reinflate to initial pressure.
- Ensure that the control knob on the pump is closed when increasing the pressure and open when decreasing the pressure.
- Ensure that the pinch clamp is open when increasing or decreasing the pressure.
- Use of the FemoStop™ Femoral Compression System is not intended to replace careful monitoring of the patient’s puncture site. The patient should not be left completely unattended during the time of compression.
- The compression system is for single use only.
- Avoid exposing the pump to any liquid.
- After use, this product may be a potential biohazard. Handle and dispose of all such devices in accordance with accepted medical practice and applicable local, state and federal laws and regulations.
- The temperature range on the label represents the temperature for long-term storage.
Adverse Events
Possible adverse effects that may result from the use of this device include but are not limited to:
- tissue necrosis
- blistering of the skin/skin abrasion
- compression injuries to nerves with subsequent sensory and motor deficits
- femoral artery and/or vein thrombosis
- embolization
- bleeding or hematoma
- arterio-venous fistula or pseudoaneurysm
- acute distension or rupture of a pseudoaneurysm during compression repair
Additional warnings and precautions for ultrasound-guided compression repair of a pseudoaneurysm in the femoral artery
Warnings
- To prevent limb ischemia, do not leave artery completely blocked for more than 3 minutes.
- Use color Doppler to periodically confirm whether or not flow remains in the vessels.
- Do not leave the system on the patient for inappropriately long compressions, as tissue damage may occur.
- During long compression periods, pressure should be briefly interrupted at least every three hours. Use manual compression during this break to limit new flow into the pseudoaneurym.
- Inappropriately long compression and/or immobilization may increase the risk for thrombosis or embolization which could lead to patient injury or death.
Precautions
- The FemoStop™ Femoral Compression System should only be used for ultrasound-guided compression repair of a pseudoaneurysm in the femoral artery, by physicians trained in the treatment of pseudoaneurysms.
- Remove any residual ultrasound gel from the skin overlying the point to be compressed as it may cause the system to slip out of position during the application of pressure.
- Exceeding pressures of 200mmHg may indicate the need to tightly secure the belt or reposition the arch.
MAT-2207286 v3.0
