
Profile and Push
The profile you need with the push you want.*
Customized engineering for multiple applications
- Offers a broad portfolio with each size range engineered for specific clinical situations
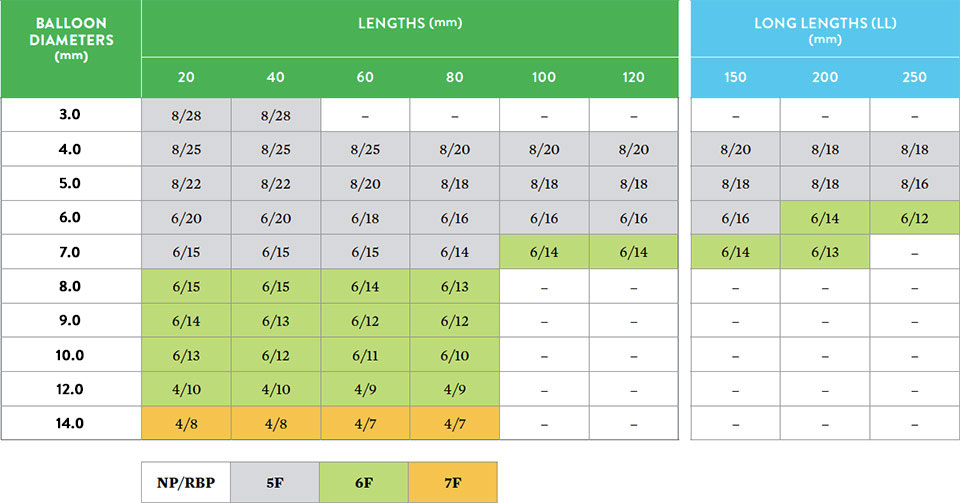
- Balloon lengths from 20 mm to 250 mm
- Balloon diameters from 3 mm to 14 mm
- Catheter lengths of 80 cm and 135 cm
- 5-7 F sheath compatibility
Outstanding performance*
- Low crossing profile and tip for ease of entering and crossing challenging lesions
- Outstanding pushability even with long lengths
- Quick deflation when it matters most
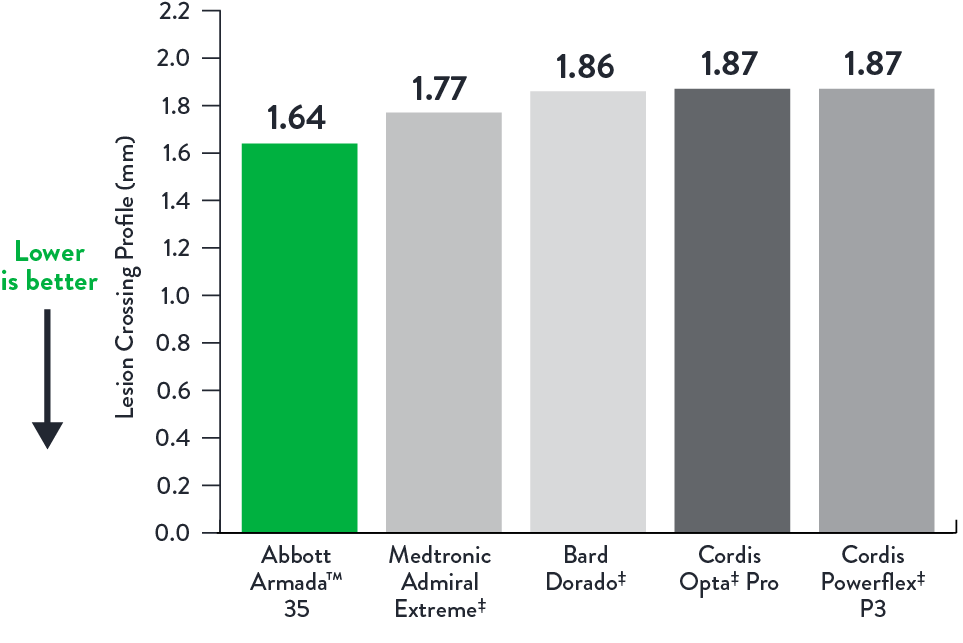
Low Crossing Profile*
Lesion Crossing Profile (mm)
Balloon Lengths 5.0 x 40 mm
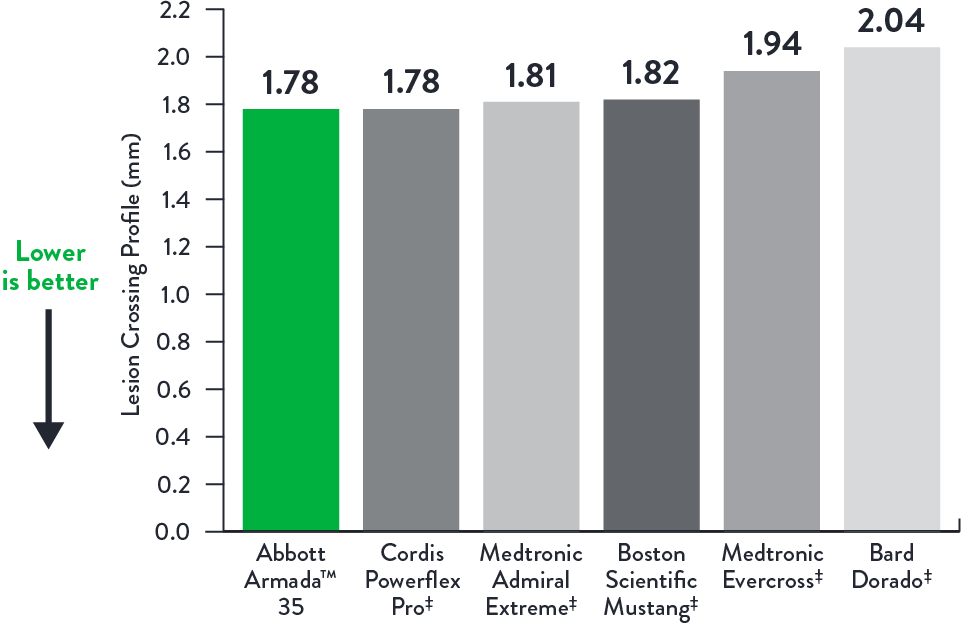
Lesion Crossing Profile (mm)
Long Balloon Lengths 6.0 x 200 mm**
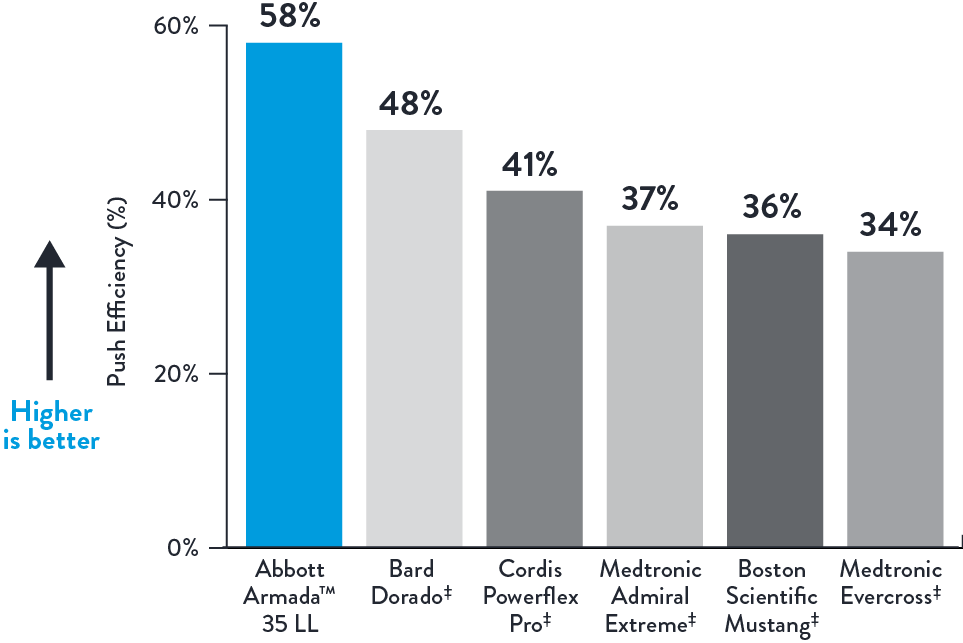
High Push Efficiency*
Push Efficiency (%)
Long Length Balloon Lengths 6.0 x 200 mm
Catheter Cross-Section
Novel crescent-shaped contrast lumen provides strong push transmission and rapid deflation.

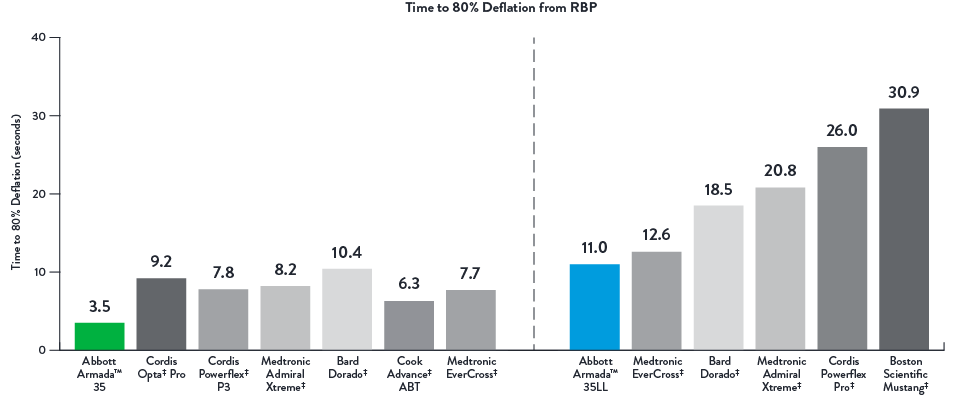
Fast Deflation Times (seconds)
Time to 80% Deflation from RBP
Balloon Compliance
Controlled three-dimensional (3D) compliance
Design, materials, and manufacturing technologies result in a low-profile balloon with controlled compliance in every dimension (i.e., diameter and length)

Balloon Compliance Across Three Dimensions (3D)
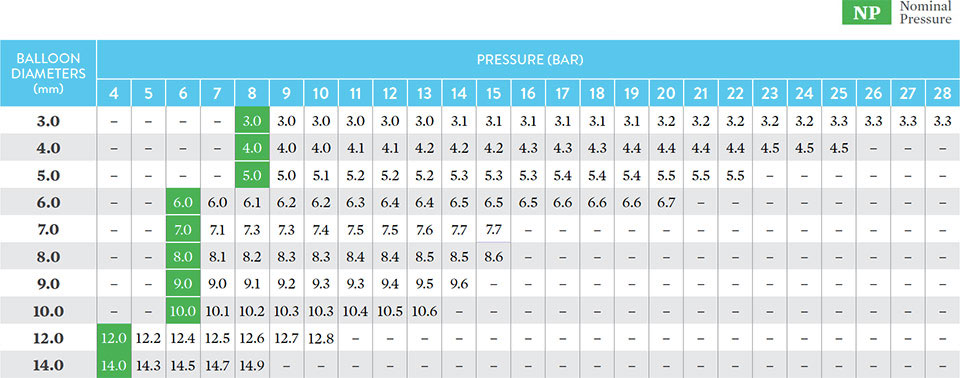
Nominal/Rated Burst Pressures and Guide Compatibility

Data on file at Abbott.
*Tests performed by and data on file at Abbott.
**All tests performed using 6x200 mm balloons except Powerflex Pro 6x220 mm.
MAT-2501199 v1.0

