Clinical Evidence
Diamondback 360 Precision™ Orbital Atherectomy System
ORBIT II Clinical Study1
The ORBIT II pivotal trial was a prospective, multi-center study conducted in the U.S. that evaluated patients with severely calcified coronary lesions treated with Diamondback 360™ Coronary Orbital Atherectomy System (OAS) prior to stent implantation.
Key Takeaways
- ORBIT II met the primary safety and efficacy endpoints by a significant margin1
- ORBIT II demonstrated long term durable results with a low target lesion revascularization (TLR) rate of 3.4% at 1 year in the drug-eluting stent (DES) subset (N=389/443)3
Study Design1
- 443 patients with severely calcified lesions were enrolled at 49 U.S. Sites
- Designed to evaluate the safety and efficacy of the Diamondback 360™ Coronary OAS
- Primary Safety Endpoint: Major adverse cardiac events (MACE) at 30 days
- Primary Efficacy Endpoint: Procedural Success defined as success in facilitating stent delivery with a residual stenosis of <50% and without the occurrence of an in-hospital MACE
PATIENT POPULATION1
| N=443 | |
|---|---|
| History of Diabetes | 36.1% |
| History of CABG | 14.7% |
| History of Dyslipidemia | 91.9% |
| History of Hypertension | 91.6% |
| Smoker (current or previous) | 66.1% |
STUDY RESULT1
| Procedural Success | 88.9% |
|---|---|
| Successful Stent Delivery | 97.7% |
| Less than 50% Residual Stenosis | 98.6% |
| Freedom from in-hospital MACE | 90.2% |
30-Day Outcomes2
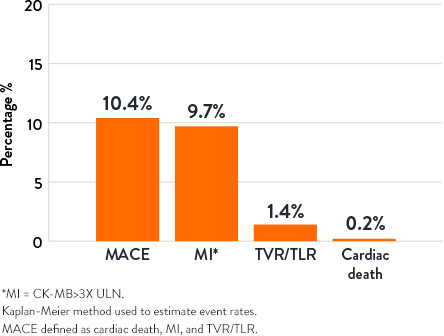
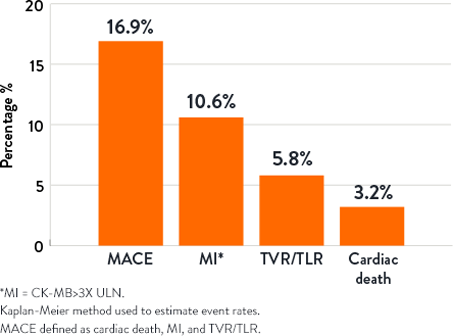
1-Year Outcomes2
- Chambers JW, et al. Pivotal trial to evaluate the safety and efficacy of the orbital atherectomy system in treating de novo, severely calcified coronary lesions (ORBIT II). JACC Cardiovasc Interv. 2014;7(5):510-8.
- Lee M, et al. Orbital atherectomy for treating de novo, severely calcified coronary lesions: 3-year results of the pivotal ORBIT II trial. Cardiovasc Revasc Med. 2017;18(4):261-264.
- Genereux P, et al. Orbital Atherectomy for Treating De Novo Severely Calcified Coronary Narrowing (1-Year Results from the Pivotal ORBIT II Trial). Am J Cardiol. 2015;115(12):1685-1690.
MAT-2310618 v2.0
Important Safety Information
Diamondback 360™ and Diamondback 360 Precision™ Coronary Orbital Atherectomy System

Including the Orbital Atherectomy Device (OAD) with GlideAssist™, Saline Pump, ViperWire Advance™ Coronary Guide Wire, and ViperWire Advance™ with Flex Tip Coronary Guide Wire
INDICATIONS
The Diamondback 360™ Coronary Orbital Atherectomy System (OAS) is a percutaneous orbital atherectomy system indicated to facilitate stent delivery in patients with coronary artery disease (CAD) who are acceptable candidates for PTCA or stenting due to de novo, severely calcified coronary artery lesions.
CONTRAINDICATIONS
Use of the OAS is contraindicated for use in the following situations:
- The ViperWire™ guide wire cannot pass across the coronary lesion.
- The target lesion is within a bypass graft or stent.
- The patient is not an appropriate candidate for bypass surgery, angioplasty, or atherectomy therapy.
- The patient has angiographic evidence of thrombus.
- The patient has only one open vessel.
- The patient has angiographic evidence of significant dissection at the treatment site.
- Women who are pregnant or children.
WARNINGS
- Do not use the OAS if the physician does not have experience in coronary angioplasty at their institution.
- Do not use the OAS if the physician does not have training on using the OAS. Contact a CSI representative for information on training.
- Do not use other commercially-available guide wires with the OAD. Only use the Model GWC-12325LG-FLP ViperWire Advance™ Coronary Guide Wire or GWC- 12325LG-FT ViperWire Advance™ Coronary Guide Wire with Flex Tip with the coronary OAD. The ViperWire™ guide wire is designed for use with all coronary OAD crown and shaft configurations.
- Never operate the OAD without normal saline and ViperSlide™ Lubricant. Continually flowing saline and ViperSlide Lubricant is required for cooling and lubricating the OAD during use in order to avoid overheating and permanent damage to the device and possible injury to the patient.
- Do not use the OAD or the ViperWire™ guide wire if their sterile package barriers are compromised or damaged.
- Do not use device during spasm of the vessel.
- Do not re-sterilize or re-use the OAD. If the OAD is re-sterilized or re-used, the OAD may not function properly potentially leading to serious infection and patient harm and/or death.
- Do not re-sterilize or re-use the ViperWire guide wire or the guide wire torquer. If the ViperWire™ guide wire or torquer is re-sterilized or re-used, the guide wire may not function properly potentially leading to serious infection and patient harm and/or death.
- Never use force to advance the spinning crown as vessel perforation may occur. If any resistance to crown travel is felt, reposition the crown away from the lesion, immediately stop treatment, and use fluoroscopy to assess the vessel for any complications. If it is confirmed there are no complications, reposition the device and advance and retract at a targeted rate of 1 to 3 mm per second.
- Use fluoroscopy to monitor and maintain spacing between the driveshaft and guide wire spring tip throughout the procedure. Always keep more than 5mm of spacing between the distal end of the OAD driveshaft and the proximal end of the guide wire spring tip. If the distance between the driveshaft tip and the ViperWire™ guide wire spring tip is insufficient, the driveshaft tip may contact the guide wire spring tip and result in dislodging the guide wire spring tip.
- Immediately stop using any OAS component should mechanical failure of any component occur before or during the atherectomy procedure. Using damaged components may result in OAS malfunction or patient injury.
- Immediately stop use of the OAD if the device stalls. Review for complications if a stall condition occurs. Do not change to high speed if device stalls on low speed.
- Note: If a stall occurs, the On/Off button is inactive for five seconds. If the On/Off button is pressed during this five second lockout period, the lockout period will begin again.
- Initial treatment for each lesion must start at low speed.
- Do not continue treatment if the wire or the device becomes subintimal.
- Do not operate the OAD if there is a bend, kink, or tight loop in the ViperWire™ guide wire. A bend, kink, or tight loop in the ViperWire™ guide wire may cause damage to and malfunctioning of the device during use.
- Performing treatment in excessively tortuous or angulated vessels or bifurcations may result in vessel damage or device failure requiring retrieval.
- Always keep the crown advancing or retracting, while it is orbiting, by continually moving the crown advancer knob to ensure corresponding (1:1) movement between the crown advancer knob and the orbiting crown.
- Do not start or stop orbiting of the crown when tight in a lesion.
- Once the OAD has reached full speed (as indicated by a stable pitch) continue to maintain a targeted travel rate of 1 to 3 mm per second, and do not exceed 10 mm per second. If the orbiting crown remains in one location it may lead to vessel damage.
- Maximum total treatment time should not exceed 5 minutes. If maximum total treatment time exceeds 5 minutes, the OAD shaft, crown, and ViperWire™ guide wire may begin to exhibit signs of wear and result in a device malfunction and possible injury to patient. A team member should track run time during use to verify total run time is not exceeded.
- Do not advance or retract the orbiting crown by advancing the OAD sheath or handle. Buckling of the ViperWire™ guide wire may occur resulting in vessel perforation or vascular trauma. Always advance the orbiting crown by using the crown advancer knob.
- Do not inject contrast solution into the OAD injection port. Device failure or patient harm may occur.
- Do not attempt aspiration through the OAD or saline line while placed within the body. If saline is pulled out through the OAD or saline line, air may enter the system.
- If air is noticed in the system while the OAD is within the body, discontinue treatment by pressing the OAS Pump power button and carefully remove the OAD driveshaft and crown from the introducer sheath/guide catheter.
- Do not allow body parts or clothing to come into contact with spinning components as the OAD orbits at very high speeds. Physical injury to the user or entanglement of clothing with the crown may occur.
- The OAS was only evaluated in severely calcified lesions; therefore the scientific evidence to support use of the OAS to treat other types of lesions/patients is limited.
- Do not spin the crown in GlideAssist™, with the guide wire brake lever in the unlocked position, without first securing the guide wire by holding it with fingers or by using the guide wire torquer. If using the guide wire torquer, ensure that it is securely fastened to the guide wire before starting to spin the crown. Failure to secure the guide wire when the brake is unlocked could allow the guide wire to spin while in GlideAssist™ mode which may result in patient harm.
PRECAUTIONS
- Do not use the OAD if there is damage to the OAD package or if the OAD has reached its shelf-life expiration date.
- If using an adjustable hemostasis valve with the guide catheter, close the hemostasis valve to minimize blood loss from around the guide catheter while still allowing the OAD sheath to slide through the hemostasis valve. Avoid excessive tightening of the hemostasis valve to prevent damaging the OAD catheter sheath. When inserting or removing the OAD crown or drive shaft through the hemostasis valve, use care not to deform the drive shaft.
- If crown and crown advancer knob movements are not moving correspondingly with one another (1:1 movement), retract and re-advance the crown into the lesion using a travel rate between 1 to 3 mm per second. Repeat retracting and advancing the crown into the lesion until crown to crown advancer knob movement correspondence is observed. If the knob and the crown are not moving together, the crown may be driven into the lesion with too much force and may result in the crown springing forward on exiting the lesion.
- Follow standard institution atherectomy policies and procedures, including those related to anticoagulation, channel blockers, and vasodilator therapy.
- Ensure fluoroscopy provides adequate visualization of the OAS system. Always use fluoroscopy to monitor the guide wire spring tip and driveshaft positions at all times throughout the procedure. If wire movement occurs, it is suggested to reposition the guide wire before advancing the device or continuing treatment.
- Applies to Diamondback 360™ Precision Coronary OAS only: Verify that contrast media injections are not above 400 psi and are not occurring during spinning of the crown.
- Applies to Diamondback 360™ Precision Coronary OAS only: If using a guide catheter smaller than 0.071 inches (1.80 mm), contrast media flow may be reduced.
- A temporary pacing lead may be necessary when treating lesions in the right coronary and circumflex arteries due to the possible occurrence of electrophysiological alternations.
- The risk of the occurrence of a dissection or perforation is increased in severely calcified lesions undergoing percutaneous treatment; therefore, on-site surgical back- up should be included as a clinical consideration. If onsite surgical back-up is not provided, then an agreement with an alternative hospital should be considered, in advance of the procedure, where the patient could be transferred in an emergency situation.
- Do not kink or crush the saline tubing as this will reduce the flow of saline and ViperSlide™ Lubricant to the OAD.
- Continually monitor and check the saline tubing and connections for leaks during the procedure.
- Do not spin the crown while advancing or retracting the crown within a guide catheter or tuohy. Damage to the guide catheter, tuohy, and/or OAD may occur.
- Ensure the OAD strain relief remains straight during atherectomy treatment. If the OAD strain relief does not remain straight, the shaft/sheath can kink.
- Do not sterilize the OAS pump. Sterilizing will damage the OAS pump. The OAS pump is intended to be used and maintained outside of the sterile field. Reference the Instructions for Use on cleaning and disinfecting the OAS pump.
- Do not allow fluid to leak onto electrical connections of the OAS pump.
- Do not spin the crown without a seated and supportive guide catheter
- When treating from a larger lumen to a smaller lumen, make sure the guide catheter is coaxial and that the tip of the OAD has entered the coronary artery to control the initial orbit before engaging the crown; engage the OAD tip into the tight stenosis until low speed has reached its treatment potential prior to initiating treatment with high speed.
- To relieve compression in the driveshaft, lock the crown advancer knob at 1cm from the full back position, advance device over wire to a position proximal from the lesion, deploy the guide wire brake, then unlock the crown advancer knob and move it fully proximal. If the OAD is started with existing compression in the driveshaft it may result in the crown springing forward.
- Do not flip contents of tray into sterile field as damage may occur. Components within tray must be carefully removed and placed into sterile field to avoid damage.
- Ejection fractions less than 25% have not been studied, use with low ejection fractions may require additional precautions due to compromised heart function.
POTENTIAL ADVERSE EVENTS
Potential adverse events that may occur and/or require intervention include, but are not limited to:
- Allergic reaction to medication/media/device components. Aneurysm. Angina (ischemic chest pain). Arrhythmias. Arteriovenous fistula. Bleeding. Bruising/hematoma. Cardiac/cardiopulmonary arrest. Cardiac/pericardial tamponade. Cerebrovascular accident (CVA). Death. Embolization, distal (air, tissue, thrombus, device). Emergent coronary artery bypass graft surgery (CABG). Failure to deliver the system to the intended locations. Fever. Heart failure/dysfunction. Hemorrhage, requiring transfusion. Hypotension/hypertension. Infection. Myocardial infarction. Pain. Pericardial effusion. Pseudoaneurysm. Restenosis of treated segment leading to revascularization. Renal insufficiency/failure. Shock (cardiogenic, hypovolemic). Slow flow or no reflow phenomenon. Stroke. Thrombus. Vessel closure, abrupt. Vessel injury, requiring surgical repair. Vessel dissection, perforation, rupture, or spasm. Vessel occlusion.
Diamondback 360™ and Diamondback 360 Precision™ Coronary Orbital Atherectomy System are manufactured and distributed by Cardiovascular Systems, Inc. (CSI). CSI is a subsidiary of the Abbott Group of Companies.
MAT-2303956 v1.0
