CardioMEMS HF System: Advanced Heart Failure Monitoring
Slow heart failure progression with early intervention using presymptomatic data
Abbott's CardioMEMS™ HF System is the leading remote hemodynamic monitoring platform for both HFpEF1 and HFrEF patients, supported by a variety of clinical trial data and proven to:
- help physicians prevent worsening heart failure2
- reduce pulmonary artery (PA) pressures1
- reduce heart failure–related hospitalizations2,3
- lower heart failure mortality rates3-5
- improve quality of life for heart failure patients6
As telemedicine becomes more common, the CardioMEMS HF System continues to be a safe, reliable way to help your patients manage their heart failure.
The CardioMEMS HF System offers you regular notification of changes in a patient’s PA pressures and simple, convenient access to secure data for proactive, personalized patient management. These insights are enabled by the CardioMEMS sensor, which measures PA pressure. The system also provides patients with a heightened awareness of the factors affecting their health and a powerful sense of control.
CardioMEMS HF System is Covered by Medicare and Medicare Advantage*

*If coverage criteria is met
Monitoring Pulmonary Artery (PA) Pressures is Proactive and Actionable
The CardioMEMS HF System remotely monitors changes in pulmonary artery pressure, an early indicator of the onset of worsening heart failure. These early indicators can often be addressed through simple adjustments to care, such as titration of medications, often without requiring an appointment with the patient.
Traditionally, clinicians have focused on physiological markers (patient weight, blood pressure, etc.) to detect worsening heart failure. Unfortunately, these markers appear late in the time course of decompensation. Relying on them leaves little time to respond before hospitalization is necessary.
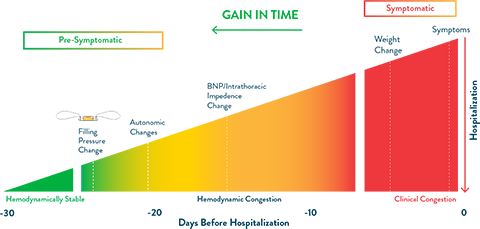
Graph adapted from Adamson PB. Pathophysiology of the transition from chronic compensated and acute decompensated heart failure: new insights from continuous monitoring devices. Current Heart Failure Reports. 2009;6:287-292.
“Before I had the CardioMEMS implanted, I was having issues around not being able to breathe."
Hear how Cynthia describes her experience with the CardioMEMS™ HF System and the difference remote pulmonary artery pressure monitoring made in her daily life.
Sign up to receive essential information and updates about hemodynamic monitoring with the CardioMems HF System
- Clinical Data
- Patient Selection
- Case Studies
- Clinican Education

The CardioMEMS HF System helps manage both HFrEF and HFpEF patients1
For patients with HFpEF, the CardioMEMS HF System helps manage symptoms and reduce hospitalizations by monitoring pulmonary artery pressures. Similarly, for those with HFrEF, the system offers critical insights into their heart function, allowing for timely interventions and optimized treatment strategies.
The CardioMEMS HF System also supports outpatient management for both HFpEF and HFrEF, enabling clinicians to make therapy adjustments through telemonitoring and emphasize early intervention in heart failure care.
Embrace the future of heart failure management with the CardioMEMS HF System, and ensure comprehensive care for HFpEF and HFrEF patients.

References
- Adamson PB, Abraham WT, Bourge RC, Costanzo MR, Hasan A, Yadav C, et al. Wireless pulmonary artery pressure monitoring guides management to reduce decompensation in heart failure with preserved ejection fraction. Circulation: Heart Failure. 2014;7(6):935-944.
- Lindenfeld J, Zile MR, Desai AS, et al. Haemodynamic-guided management of heart failure (GUIDE-HF): a randomized controlled trial. The Lancet. 2021;398:991-1001.
- Abraham J, Bharmi R, Jonsson O, et al. Association of ambulatory hemodynamic monitoring with clinical outcomes in a concurrent matched cohort analysis. JAMA Cardiology. 2019;4(6):556-563.
- Givertz MM, Stevenson LW, Costanzo MR, et al., on behalf of the CHAMPION Trial Investigators. Pulmonary artery pressure–guided management of patients with heart failure and reduced ejection fraction. J Am Coll Cardiol. 2017;70:1875-86.
- Lindenfeld, J, Costanzo, MR, Zile, MR, on behalf of the GUIDE-HF, CHAMPION, and LAPTOP-HF investigators. Implantable Hemodynamic Monitors Improve Survival in Patients with Heart Failure and Reduced Ejection Fraction. Journal of the American College of Cardiology (JACC). 2024;83(6):682-694.
- Brugts, J et al. Remote haemodynamic monitoring of pulmonary artery pressures in patients with chronic heart failure (MONITOR-HF): a randomised clinical trial. The Lancet. May 20, 2023. https://doi.org/10.1016/S0140-6736(23)00923-6
MAT-2006906 v12.0
Indications, Safety & Warnings
Rx Only
Brief Summary: Prior to using these devices, please review the Instructions for Use for a complete listing of indications, contraindications, warnings, precautions, potential adverse events and directions for use.
CardioMEMS™ HF System Indications and Usage: The CardioMEMS HF System is indicated for wirelessly measuring and monitoring pulmonary artery pressure and heart rate in NYHA Class II or III heart failure patients who either have been hospitalized for heart failure in the previous year and/or have elevated natriuretic peptides. The hemodynamic data are used by physicians for heart failure management with the goal of controlling pulmonary artery pressures and reducing heart failure hospitalizations.
CardioMEMS HF System Contraindications: The CardioMEMS HF System is contraindicated for patients with an inability to take dual antiplatelet or anticoagulants for one month post implant.
CardioMEMS HF System Adverse Events: Potential adverse events associated with the implantation procedure include, but are not limited to, the following: air embolism, allergic reaction, infection, delayed wound healing, arrhythmias, bleeding, hemoptysis, hematoma, nausea, cerebrovascular accident, thrombus, cardiovascular injury, myocardial infarction, death, embolism, thermal burn, cardiac perforation, pneumothorax, thoracic duct injury or hemothorax.
MAT-2006766 v7.0
