Clinical Evidence
Clinical Trials
PEVAR Clinical Trial
Objective: To compare the safety and effectiveness of the 'pre-close' technique for percutaneous femoral artery (FA) access and closure (PEVAR) to Surgical Cutdown (SEVAR).
Key Findings
- Mean procedure time was reduced in PEVAR patients by 34 minutes (106.5 ± 44.9 minutes PEVAR vs. 141.1 ± 73.4 minutes SEVAR, p=.0056)1
- Mean time to hemostasis was reduced following PEVAR by 13 minutes (9.8 ± 17.0 minutes PEVAR vs. 22.7 ± 22.9 minutes SEVAR, p=.0023)1
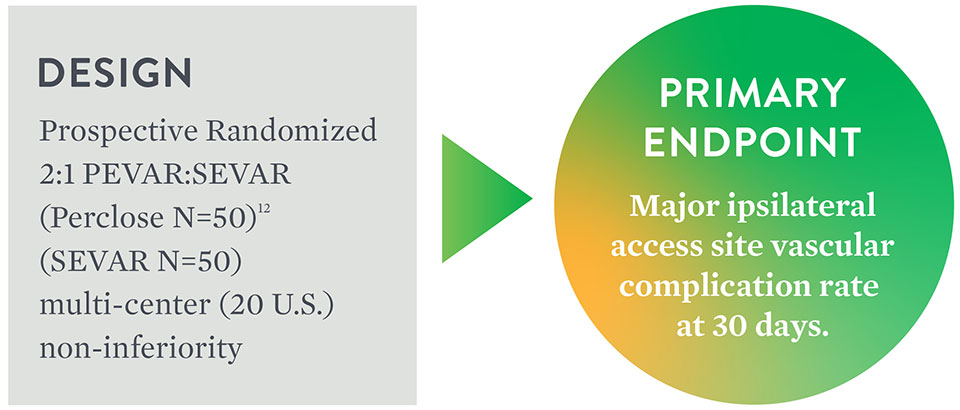
Primary Endpoint Achieved
Major6 Ipsilateral Vascular Access
Complications at 30 Days
| PEVAR Perclose N=50 | SEVAR N=50 | Difference 95% CI3 | p-value3 | |
|---|---|---|---|---|
| Major Complications [95% CI]7 | 6% (3/50) [1.3%, 16.5%] | 10% (5/50) [3.3%, 21.8%] | -4.0% [-, 4.9%] | 0.0048 |
Composite of Minor8 Ipsilateral Vascular Access
Complications at 30 Days
| PEVAR Perclose N=50 | SEVAR N=50 | p-value3 | |
|---|---|---|---|
| Minor Ipsilateral Access Site Vascular Complications at 30 Days [95% CI]7 | 4% (2/50) [0.5%, 13.7%] | 8% (4/50) [2.2%, 19.2%] | 0.67774 |
1. By two-sample t-test.
2. PEVAR-Perclose analyzed independently from Prostar™ XL.
3. One-sided p-value and 95% confidence interval for non-inferiority test by using asymptotic test statistics with non-inferiority margin of 10%.
4. No statistically significant difference.
5. By Fisher's exact test.
6. Major Complications: access site vascular injury requiring repair; new onset lower extremity ischemia requiring surgical or percutaneous intervention; Access site-related bleeding requiring transfusion; access site-related infection requiring IV antibiotic or prolonged hospitalization; access site-related nerve injury that is permanent or requires surgery.
7. By Clopper-Pearson exact confidence interval.
8. Minor Complications: pseudoaneurysm or AV fistula; hematoma ≥ 6 cm; post-discharge bleeding requiring > 30 minutes to re-achieve hemostasis; lower extremity arterial emboli or stenosis attributed to access site; deep vein thrombosis; access site-related vascular laceration; transient access site-related nerve injury; access site wound dehiscence; access site-related lymphocele; localized access site infection treated with IM or PO antibiotic.
Source: Nelson, Peter R. et al. A multicenter, randomized, controlled trial of totally percutaneous access versus open femoral exposure for endovascular aortic aneurysm repair (the PEVAR trial). Journal of Vascular Surgery. (2014): 1181-1193.
Clinical Evidence on Closure of Large-Bore Venous Access
Perclose Cohort in the REALISM9 Clinical Trial
A prospective analysis was performed to evaluate the safety and effectiveness of the Perclose in closing large-sized venous access sites through a retrospective data collection. The prospective analysis included subjects in whom Perclose was used as the primary method for large-bore venous access site closure during the TMVr index procedure with a 24F vascular sheath.
Key Findings
- Major complication was low at 1.9%
- Freedom from major femoral vein access site-related complications was 98.1% at 30 days
- Perclose is safe and effective in the closure of venous access-site with up to 24F sheath
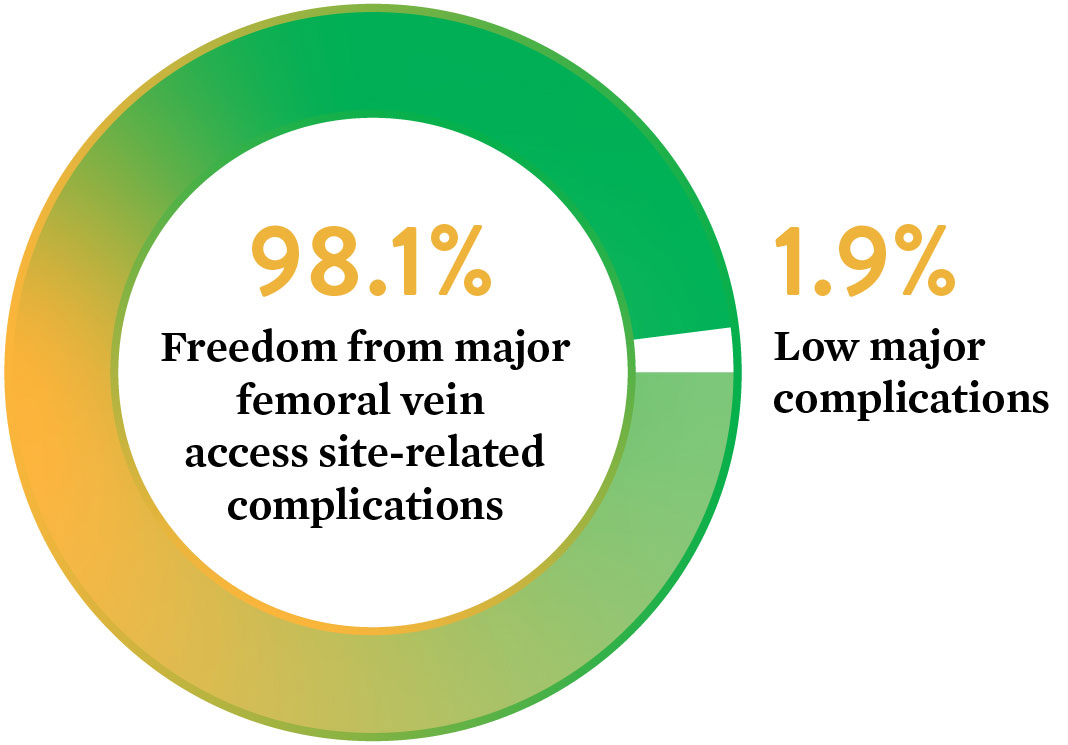
Low major complications at 30 days
9. EVEREST II/REALISM Continued Access Registry Study.
Source: The Use of the Perclose ProGlide™ Suture Mediated Closure (SMC) Device for Venous Access-Site Closure up to 24F Sheaths. Kar, Saibal; Hermiller, James; et al. CRT 2018.
Real-World Evidence on Repair of Large-Bore Arterial Access
Perclose vs. Surgical Cutdown
The Perclose vs. Surgical Cutdown retrospective study is designed to compare clinical outcomes and complication rates among patients undergoing closure of large-bore arterial access using Perclose versus Surgical Cutdown (Cutdown) in a real-world setting.
Key Findings
The use of Perclose for repair of large-bore arterial access is associated with significantly lower blood transfusions, infections, mortality, and length of stay compared to Surgical Cutdown.
Patient Baseline
| Cutdown | Perclose | |
|---|---|---|
| # of Patients | 757 | 757 |
| Anticoagulants◊ | 17.8% | 44.9% |
◊p<0.05
Patients may have had multiple procedures during index admission
Perclose Patients
Less likely to have an infection
Shorter hospital stay
Less likely to die within 30 days post-procedure
Less likely to require a blood transfusion
Source: Perclose ProGlide™ Versus Surgical Closure Outcomes – Real World Evidence. Schneider, Darren B; Krajcer, Zvonimir; et al. LINC 2018.
PEVAR PUBLICATIONS
Perclose™ ProStyle™ SMCR System is a design evolution of the Perclose ProGlide™ SMC System. The results in the following PEVAR publications are applicable to the Perclose™ ProStyle™ SMCR System because of system similarities.
Italian Percutaneous EVAR (IPER) Registry
Outcomes of 2,381 percutaneous femoral access sites’ closure for aortic stent-graft
Objective: The aim of this publication was to report outcomes of endovascular aneurysm repair with percutaneous femoral access (PEVAR) using Prostar™ XL and Perclose closure systems, from the multi-center Italian Percutaneous EVAR (IPER) registry.
Method
- Prospective
– 7 centers
– Operators with at least 50 closure device deployments - January 2010 – December 2014
– Use of Perclose began in 2013 - 2,381 femoral access sites
– 514 Perclose closes
– 1,867 Prostar closes - > 18F sheath size: 26.5%
Perclose-Only Result
- 97.5% (501/514) technical success rate10
Total Results including Prostar
- 3.2% (76/2,381) major complications11 requiring surgical cutdown
- 1.1% (27/2,381) minor complication12
- 0.25% (6/2,381) puncture site complication observed during one-month follow-up
Key Takeaways
- High device success (Perclose 97.5%) even with demanding cases due to the presence of obesity (24.7%), femoral artery calcification (53%), and iliac tortuosity (22.6%)
- First and largest prospective study carried out on a cohort of unselected patients undergoing PEVAR with different endografts in well-trained centers by highly experienced operators
- Femoral calcification was the only independent predictor of technical failure
10. Technical success was defined as the ability to obtain a successful percutaneous vascular access and closure without serious complications.
11. Major access-related complications included all the complications requiring an early surgical conversion due to bleeding, pseudoaneurysm, CFA stenosis/occlusion, and were considered a technical failure of system closure.
12. Minor access-related complications were related to the presence of minimal bleeding at the access site not requiring surgical conversion.
Source: Pratesi, G., et al. "Italian Percutaneous EVAR (IPER) Registry: outcomes of 2,381 percutaneous femoral access sites' closure for aortic stent-graft." The Journal of Cardiovascular Surgery 56.6 (2015): 889-898.
Perclose in PEVAR and TAVI – Brazil Study
Objective: Assessment of efficacy, complication, and potential risk factors for Perclose
Method
- 123 Patients
– 47.6% TAVI – 33.5% EVAR
– 13.7% TEVAR, 3.2% TAAA - 242 vascular access sites
- Single-center retrospective study
- ≥ 18F sheath size: 54.3%
- Single operator with 50 small-hole closure experience
(no prior large-hole experience) - Angiotomography instead of ultrasound used for puncture
Perclose Result
- 98.37% (121/123) patient technical success rate13
- 1.62% acute major complication rate
– Failures had > 2/3 CFA calcification - 0.81% (1/123) minor complication observed at 30-day follow-up
- Operator encountered an initial learning curve with the 3 complications occurring during the first 35 cases
Key Takeaways
- Experience with Perclose for small-hole closure helps with large-hole closure success
- Pre-operative imaging on the puncture site to assess calcification important for device success
13. Device failure defined as failure to achieve hemostasis at the access site, leading to alternative treatment other than manual compression.
Source: Saadi, Eduardo Keller, et al. "Totally Percutaneous Access Using Perclose ProGlide™ for Endovascular Treatment of Aortic Diseases." Brazilian Journal of Cardiovascular Surgery 32.1 (2017): 43-48.
Perclose in PEVAR – Taiwan Study
Objective: Evaluate the outcomes and predictive factors for additional Perclose deployment
Method
- 268 patients, 418 vascular access sites
- 9 operators
– 3 were within the learning curve for Perclose14 - Factors measured
– Depth
– Sheath size
– CFA diameter
– Any CFA calcification- Anterior wall
- Lateral wall
- Medial wall
- Posterior wall
Perclose Result
- 87.6% (366/418) Primary technical success15 with two Perclose
- 99% secondary technical success16 with additional Perclose device
– 38 required one additional device and 10 required two additional devices - Only anterior CFA wall calcification was a significant predictor for additional Perclose deployment
Key Takeaways
- It is important to perform a femoral angiogram to evaluate the severity and location of calcification at the puncture site
- Even with additional device deployment, high success rate was able to be achieved even in the presence of anterior wall calcification (17% of total access sites)
14. Learning curve shown to be most pronounced over first 18 months. Bechara C.F. et al.: Predicting the learning curve and failures of total percutaneous endovascular aortic aneurysm repair. J Vasc Surg 2013; 57: pp. 72-76.
15. Primary technical success rate defined as the ability to achieve hemostasis without additional ProGlide™ device deployment or surgical repair.
16. Secondary technical success rate defined as the ability to achieve hemostasis after deployment of one or two ProGlide™ devices without surgical repair.
Source: Lin, Shen-Yen, et al. "Predictive Factors for Additional Perclose Deployment in Percutaneous Endovascular Aortic Repair." Journal of Vascular and Interventional Radiology 28.4 (2017): 570-575.
Perclose LIFE Registry
Objective: Demonstrate clinical and cost benefits when utilizing a Fast-Track EVAR protocol with the Ovation stent graft system
Method
- Prospective multi-center registry
- 250 patients, 35 U.S. centers
- 14F Ovation Abdominal Stent Graft
- Fast-Track EVAR Protocol
– Percutaneous access
– No general anesthesia
– No ICU admission
– Next-day discharge
Perclose Result
- 97.6% Perclose technical success17 rate (244/250)
- No device-related or procedure-related major adverse events
- Cost analysis shows Fast-Track EVAR is less costly than standard EVAR
Cost Analysis
| Fast Track | Standard EVAR*** | Fast-Track Savings | |
|---|---|---|---|
| Anesthesia | Local/Regional $300 | General $500 | $200 |
| Access | Bilateral PEVAR $1,200 | Cutdown† $300 | ($900) |
| ICU | 0% $0 | 1.4 Days, 51% $15,300 | $15,300 |
| Non-ICU | 1.2 Days $6,700 | 2.3 Days $12,900 | $6,200 |
| 30D Reintervention | 0% $0 | $29.4K, 1.1% $300 | $300 |
| Total | $8,200 | $29,300 | $21,100 |
**Standard EVAR Control group: Benchmarked 8,306 patients treated with standard, elective infrarenal EVAR at an alliance of ~3,750 U.S. Premier hospital facilities, based on Inpatient Discharge between 2012-2015.
***Standard EVAR: Average costs per patient, extracted costs for access, anesthesia, ICU, and hospital stay to calculate costs associated with Fast-Track.
†Assumes 30% applicability based on anatomic criteria, with 23% bilateral PEVAR and 7.0% unilateral PEVAR (Manunga et al., J Vasc Surg. 2013).
Source: Kracjcer, Z. Fast-Track Endovascular Aortic Repair: Final Results from the Prospective LIFE registry. VIVA 2016.
Key Takeaways
- Fast-Track is less costly than standard EVAR with hospital and ICU stay as the main cost drivers
- Patient selection is critical to success as femoral arteries should be free of heavy calcification or extreme tortuosity to facilitate bilateral percutaneous access
17. Technical Success (Successful Bilateral PEVAR)
Source: Kracjcer, Z. Fast-Track Endovascular Aortic Repair: Final Results from the Prospective LIFE registry. VIVA 2016.
TAVI Publications
Perclose™ ProStyle™ SMCR System is a design evolution of the Perclose ProGlide™ SMC System. The results in the following TAVI publications are applicable to the Perclose™ ProStyle™ SMCR System because of system similarities.
Perclose in TAVI – CONTROL Study
Objective: To evaluate the efficacy of Perclose
Method
- Retrospective study
- 9 centers in Europe, North America, and the Middle East
- 472 Perclose compared to 472 Prostar deployment
- TAVI devices used: Sapien (51.1%) and CoreValve (43.2%)
- 18.3 ± 1.7F mean sheath size
Key Takeaways
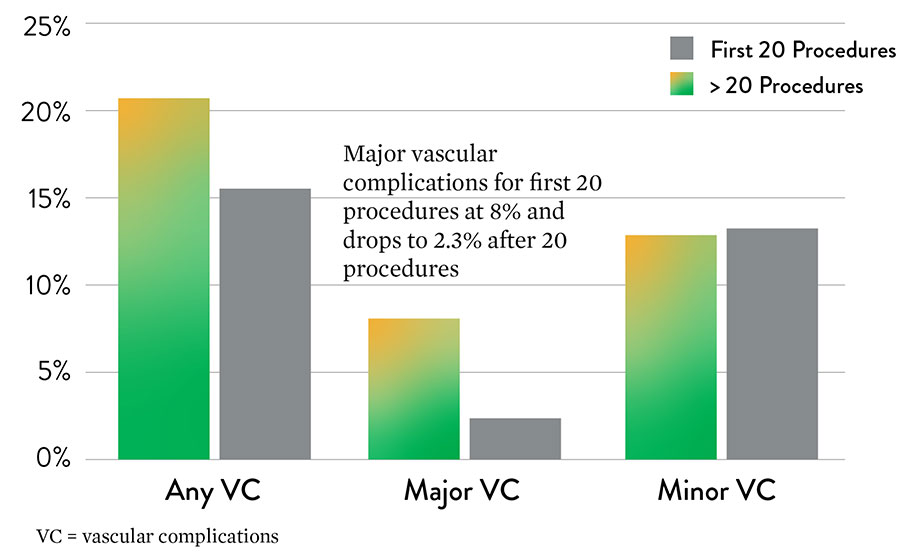
- Low major vascular complication rate in TAVI procedures – 1.9%
- Major complication rate decreased significantly after first 20 procedures, showing a learning curve effect
Source: Barbash, Israel M., et al. "Comparison of vascular closure devices for access site closure after transfemoral aortic valve implantation." European Heart Journal 36.47 (2015): 3370-3379.
Perclose-Only Result
- 1.9% (9/472) major complications
- 18% (85/472) minor complications
- There was a significant decrease in major vascular complications after the first 20 cases, showing a learning curve effect
- Major vascular complications consisted of rupture, dissection, perforation, access site hematoma and pseudoaneurysm formation requiring urgent vascular surgery. Minor complications resulted in unplanned balloon angioplasty, stenting, and the need for unplanned surgical intervention.
Perclose in TAVI – Germany Study
Objective: Evaluate the safety and efficacy of Perclose
Method
- Prospective single-center study
- 348 Perclose patients compared to 237 Prostar patients
- TAVI devices used
– Sapien 3
– Lotus
– CoreValve - Sheath size 23.7F ± 1.5F
Perclose-Only Result
- 95.4% (332/348) technical success rate18
- 2.3% (8/348) major vascular complications
- 5.7% (20/472) minor vascular complications
- Major vascular complications consisted of major dissections at the access site; and minor vascular complications consisted of leakage, dissection, or occlusion requiring unplanned stenting, surgical repair, or a balloon at the access site.
Key Takeaways
- High technical success rate
- Low major vascular complication in TAVI procedures
18. Device failure defined as failure to achieve hemostasis at the access site leading to alternative treatment other than manual compression. 4.6% (n=16) needed stenting for device failure.
Source: Seeger, Julia, et al. “Impact of suture mediated femoral access site closure with the Prostar XL compared to the Perclose system on outcome in transfemoral aortic valve implantation.” International Journal of Cardiology 223 (2016): 564-567.
Perclose in PEVAR and TAVI – Brazil Study
Objective: Assessment of efficacy, complication, and potential risk factors for Perclose
Method
- 123 Patients
– 47.6% TAVI
– 33.5% EVAR
– 13.7% TEVAR, 3.2% TAAA - 242 vascular access sites
- Single-center retrospective study
- ≥ 18F sheath size: 54.3%
- Single operator with 50 small-hole closure experience (no prior large-hole experience)
- Angiotomography instead of ultrasound used for puncture
Perclose-Only Result
- 98.37% (121/123) patient technical success rate19
- 1.62% acute major complication rate
– Failures had > 2/3 CFA calcification - 0.81% (1/123) minor complication observed at 30-day follow-up
- Operator encountered an initial learning curve with the 3 complications occurring during the first 35 cases
Key Takeaways
- Experience with Perclose for small-hole closure helps with large-hole closure success
- Pre-operative imaging on the puncture site to assess calcification important for device success
19. Device failure defined as failure to achieve hemostasis at the access site, leading to alternative treatment other than manual compression.
Source: Saadi, Eduardo Keller, et al. "Totally Percutaneous Access Using Perclose ProGlide™ for Endovascular Treatment of Aortic Diseases." Brazilian Journal of Cardiovascular Surgery 32.1 (2017): 43-48.
TMVr Publications
Perclose™ ProStyle™ SMCR System is a design evolution of the Perclose ProGlide™ SMC System. The results of the REALISM Clinical Trial are applicable to the Perclose ProStyle SMCR System because of system similarities.
Prospective Analysis for Large-Bore Venous Access
The Use of the Perclose ProGlide™ Suture-Mediated Closure (SMC) Device for Venous Access Site
Safety and performance of Perclose ProGlide™ vascular closure device in managing large-hole venous access site
Objective: The primary objective of this study was to evaluate the safety and performance of ProGlide™ in the closure of the venous access site in subjects treated with a large-caliber femoral vein sheath (24F).
Early Ambulation and Discharge
AF Ablation
Time management, patient and staff flow, hospital costs, and throughput can all impact overall hospital efficiency. Access site management, early ambulation and discharge with Perclose ProGlide™ Suture-Mediated Closure System can help reduce patient length of stay, optimize nursing time, improve patient flow, and drive overall hospital efficiency.
Early Ambulation and Same-Day Discharge for Atrial Fibrillation Ablation Cases
Dr. Sumit Verma from Baptist Heart and Vascular Institute discusses how the facility implemented same-day discharge for atrial fibrillation ablation cases and how the strategy has the potential to reduce complications, decrease resource utilization, and allow cost savings, while improving patient satisfaction.

MAT-2103414 v2.0
