How CardioMEMS HF System Remote PA Pressure Monitoring Works
The CardioMEMS™ HF System provides pulmonary artery (PA) pressure remote monitoring using a small sensor. Permanently implanted in the distal pulmonary artery via a safe right heart catheterization procedure, the sensor measures changes in pulmonary artery pressure. These changes are a surrogate measure for fluid retention in the lungs caused by worsening heart failure.
Patient-initiated sensor readings are wirelessly transmitted to a secure website for clinicians to access and review. Directly monitoring PA pressure not only alerts you if a patient’s heart failure is worsening, it also allows you to intervene earlier, adjusting medication or making other treatment changes, often before a patient experiences any symptoms.
The CardioMEMS HF System Sends PA Pressure Data
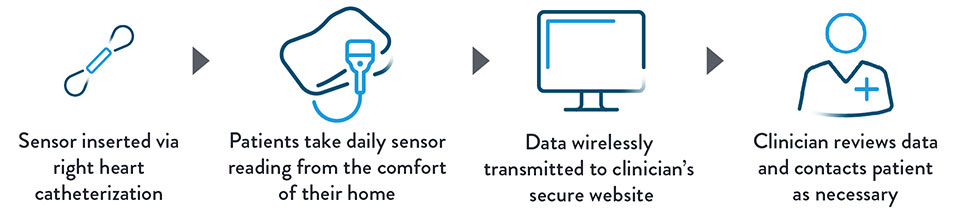
- The PA sensor is inserted via right heart catheterization
- Patient takes daily sensor readings using the portable CardioMEMS™ Patient Electronics System
- Data wirelessly transmitted to Merlin.net™ Patient Care Network, a secure website that easily presents PA pressure data to inform proactive treatment modifications
- Clinician reviews data and contacts patient, as necessary
Merlin.net Patient Care Network (PCN) Remote Monitoring Platform
The Merlin.net Patient Care Network (PCN) is a remote monitoring platform that presents pulmonary artery (PA) pressure data as actionable information. This information can guide earlier, more appropriate intervention for each patient.
The platform incorporates remote PA pressure data with Implanted Electronic Device (IED) diagnostics, such as:
- AT/AF burden
- Percent ventricular pacing
- Patient activity
- Day and night heart rate
- VT/VF events with antitachycardia (ATP) and shock therapies
This device is commercially available for use in select international markets.
MAT-2116106 v3.0
