How It Works
The system is made up of three groups of components:
- EnSite™ Amplifier
- Display workstation
- EnSite Precision™ Module
Software
The system includes the EnSite™ Courier™ PACs Module v3.0, EnSite™ Derexi™ Module v.1.3.1, EnSite™ Verismo™ Module v.2.0.1 and EnSite™ Fusion™ Module v.7.1.
Hardware
- The EnSite Precision™ Field Frame universally mounts underneath the patient bed and generates a low-powered magnetic field within which the position of a Sensor Enabled™ device can be detected.
- The EnSite Precision™ Link, Sensor Enabled™ connects Sensor Enabled tools, including catheters, patient reference sensors (PRSs) and the field frame and relays the information to the EnSite amplifier via fiber-optic connection.
Disposables
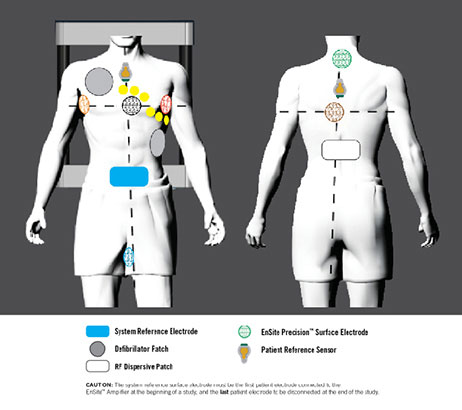
The EnSite Precision™ surface electrode kit consists of a system reference surface electrode and six surface electrodes placed on the patient in pairs: anterior to posterior, left to right lateral and superior (neck) to inferior (leg) (Figure 1).
The three electrode pairs form three orthogonal axes (X-Y-Z), with the heart at the center. Two PRSs are also used with the EnSite Precision Cardiac Mapping System: a PRS anterior, and a PRS posterior. The PRSs are connected to the patient using disposable, self-adhesive patches, and primarily function as sensors for metal distortion and patient movement.
During model collection, both impedance-based points and magnetic-based points are collected from a Sensor Enabled tool.
Impedance Data
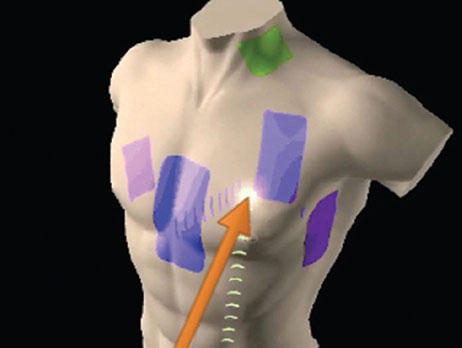
Catheter location and navigation of all compatible tools, both conventional and Sensor Enabled, are based on the impedance field generated by the EnSite surface electrodes. When the surface electrodes are connected to the EnSite Precision Cardiac Mapping System, an 8 kHz signal is sent alternately through each pair of surface electrodes to create a voltage gradient along each axis, forming a transthoracic electrical field.
Conventional or Sensor Enabled electrophysiology catheters are connected to the EnSite Precision Cardiac Mapping System and advanced to the heart. As a catheter enters the transthoracic field, each catheter electrode senses voltage, timed to the creation of the gradient along each axis (Figure 2).
Using the sensed voltages compared to the voltage gradient on all three axes, the EnSite Precision Cardiac Mapping System calculates the three-dimensional position of each catheter electrode for all electrodes simultaneously.
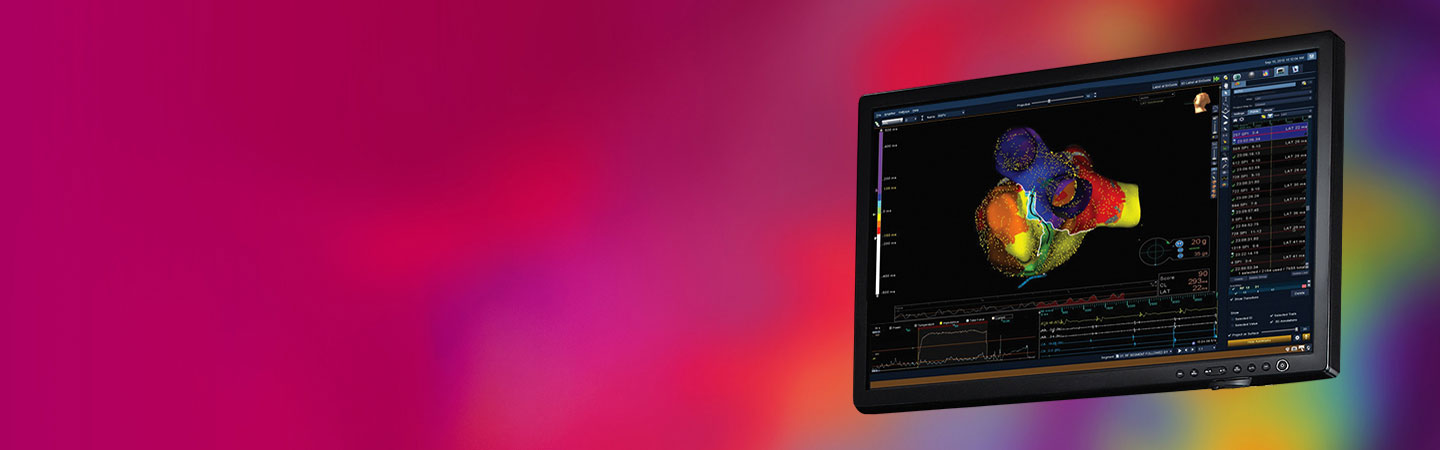
The EnSite Precision Cardiac Mapping System displays the located electrodes as catheter bodies with real-time navigation. It permits the simultaneous display of multiple catheter electrodes (Figure 3) and also reflects real-time motion of ablation and diagnostic catheters in the heart.3 By tracking the position of the catheters, the system enables the creation of 3-D electroanatomical models of the cardiac chambers.
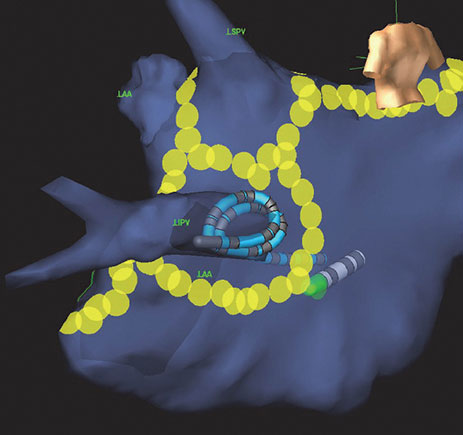
Magnetic Data
When a Sensor Enabled catheter is introduced and EnSite™ NavX™ Navigation and Visualization Technology, Sensor Enabled Field Scaling is applied, the EnSite Precision Cardiac Mapping System dynamically optimizes the model by adjusting the dimensions of the navigation field using known offsets between the position and orientation of magnetic sensor(s) and electrodes.
The EnSite Precision Cardiac Mapping System also uses magnetic information as an input for the EnGuide stability monitor to monitor field stability for unexpected changes. This feature can be enabled and used to monitor the location of a Sensor Enabled tool real time within an EnSite NavX Navigation and Visualization Technology, Sensor Enabled field scaled mode.
Intuitive Automation1
With the EnSite Precision Cardiac Mapping System, you can decrease mapping time using intelligent automation tools.1*
- Enhance VT mapping with automated, advanced morphology matching capability1
- Automatically reject catheter ectopy
- Increase procedural consistency through automated guidance of lesion marking using the AutoMark feature4
Superior Flexibility1**
The EnSite Precision Cardiac Mapping System helps you customize your procedure to address the circumstance of each case.
- Map any chamber with any catheter.***
- Choose the workflow you need to accommodate patient needs.
- Use the system to optimally integrate magnetic and impedance data.
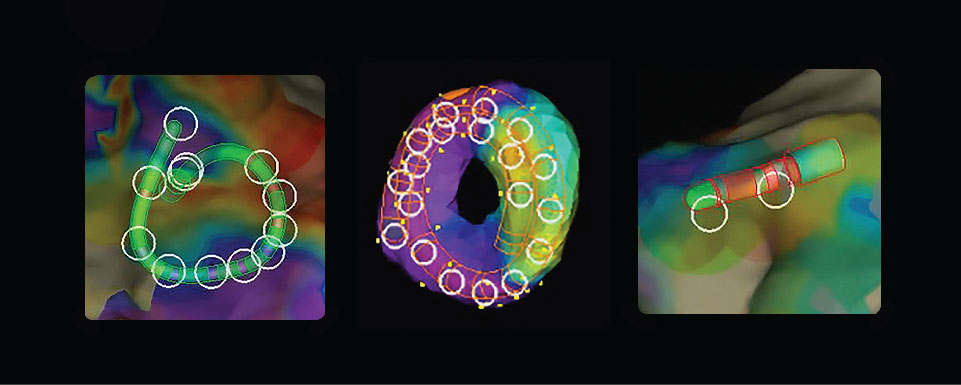
Create Faster High-Density Maps Using Any Catheter1***
Greater Precision2****
The EnSite Precision Cardiac Mapping System combines impedance and magnetics with a suite of innovative tools to give you comprehensive patient information.
- Enhance model creation with both impedance-field flexibility and magnetic-field stability.2
- Improve stability with redesigned patches that offer5:
- Improved hydrogel adhesive5
- Improved ECG patch placement options
- More accommodation for patients of all sizes
- Maintain a seamless workflow, now with greater precision via an automated sheath filter.4
3-D Models With CT-Scan-Like Detail—27x† Higher Point Density4,6
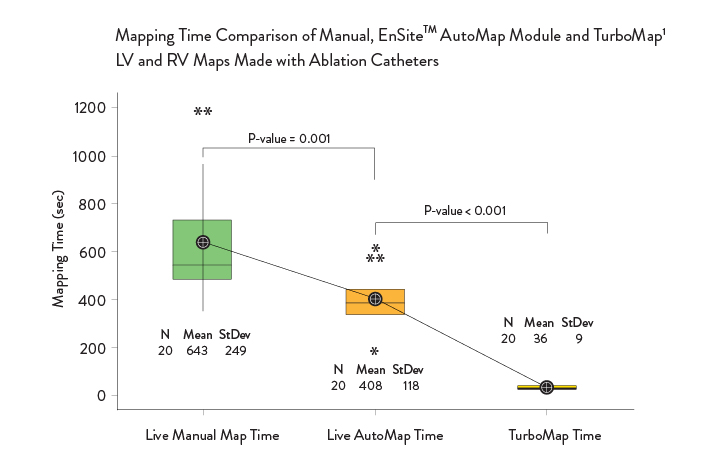
Improve Mapping Time1*
With EnSite Precision Cardiac Mapping System, you can:
- Map secondary arrhythmias up to 10x faster with the TurboMap feature1
- Have access to both positive and negative morphology score matching that may accentuate scar regions and aid in visualization of critical isthmus channels1
- Create faster, more accurate maps with greater consistency across cases1
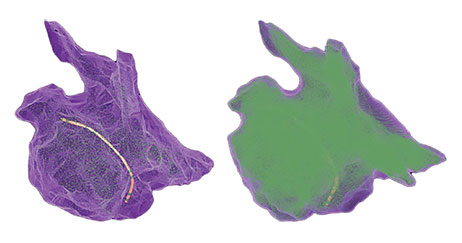
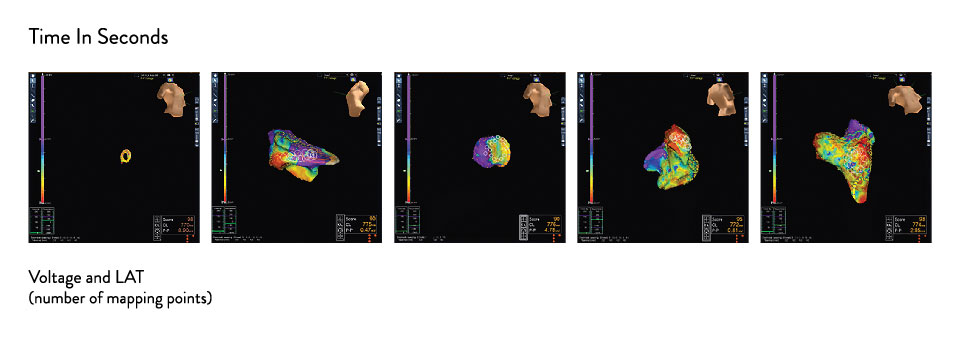
Use Onemodel and Onemap with the EnSite Automap Module
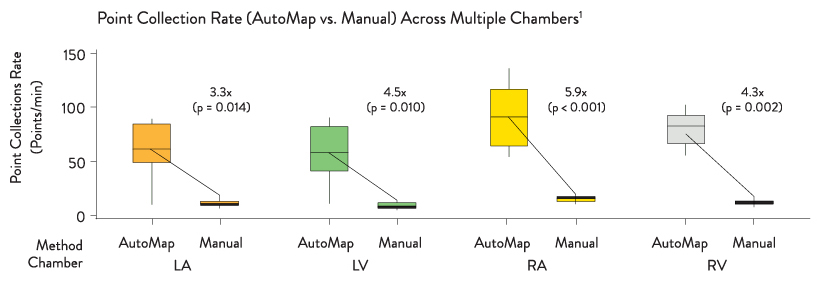
The OneMap and OneModel tools in the EnSite Precision Cardiac Mapping System can be used alongside the EnSite™ AutoMap Module for enhanced mapping capability. The EnSite Precision Cardiac Mapping System with the EnSite AutoMap Module lets you:
- Achieve 54% faster, real-time model creation with the OneModel tool — offering precise anatomic modeling7,8
- Gain visibility into a patient’s rhythm in the fewest possible cardiac cycles when using the EnSite AutoMap module with the OneMap tool — providing simultaneous collection of anatomic and electrical points from multiple electrodes8
- Create higher density map in less time — automatically with any catheter1***
This device is commercially available for use in select international markets.
* Comparison is versus manual mapping with EnSite Precision mapping module or manual lesion marking.
** The open-platform feature of the EnSite Precision™ cardiac mapping system allows for use of almost any catheter for mapping, thus offering superior flexibility as compared to the CARTO‡ system by Biosense Webster, which limits use to Biosense Webster catheters only.
*** In accordance with catheter indication for use.
**** Greater precision based on improvement in accuracy of impedance model with magnetic field scaling applied via robot testing vs. EnSite Velocity™ v4.0.2.
References
- Ptaszek LM, Moon B, Rozen G, Mahapatra S, Mansour M. (2018). Novel automated point collection software facilitates rapid, high-density electroanatomic mapping with multiple catheter types. J Cardiovasc Electrophysiol, 29(1), 186-195. https://doi.org/10.1111/jce.13368.
- St. Jude Medical. Data on file, Report 90237452.
- Packer, D. L. (2005). Three-dimensional mapping in interventional electrophysiology: techniques and technology. Journal of Cardiovascular Electrophysiology, 16(10), 1110-1116.
- St. Jude Medical. Data on file, Report 90214738.
- St. Jude Medical. Data on file, Report 90213771.
- Heist, E. K., Perna, F., Chalhoub, F., Danik, S., Barrett, C., Houghtaling, C., … Mansour, M. (2013). Comparison of electroanatomical mapping system: Accuracy in left atrial mapping. Pacing and Clinical Electrophysiology, 36(626-631). https://dx.doi.org/10.1111/pace.12095
- St. Jude Medical. Data on file, Report 90114565.
- Heist, E. K., Danik, S., Chalhoub, F., Koci, F., Barrett, C., Perna, F., … Mansour, M. (2011). Human and animal feasibility study of investigational 3D geometry acquisition software. Heart Rhythm, 8, S241, https://dx.doi.org/10.1016/j.hrthm.2011.03.027
MAT-2512390 v1.0