Ablation Technology
Ablation procedures are often long and complex. You need a catheter that offers reliability and comfortable usability, and helps you achieve successful clinical outcomes. We offer innovations in ablation technologies that support your needs and are tailored for differences in your patients’ anatomies and conditions - from RF to PFA.
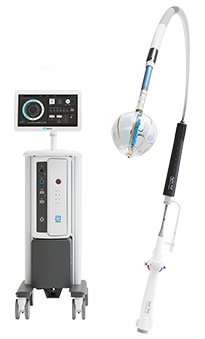
Volt PFA System
Volt™ PFA System simplifies therapy delivery, minimizing procedural burden so you can treat more patients with ease and precision1*
TactiFlex Ablation Catheter, Sensor Enabled
The first and only contact force catheter with a flexible tip, TactiFlex™ Ablation Catheter, Sensor Enabled™ was designed for optimal safety and stability, confident lesion creation and procedural efficiency, and offers full integration with the revolutionary EnSite™ X EP System.2
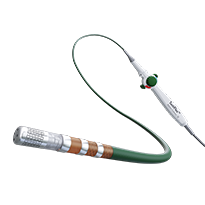
TactiCath Contact Force Ablation Catheter, Sensor Enabled
TactiCath™ Contact Force Ablation Catheter, Sensor Enabled™ features advanced handle-shaft technology for maneuverability, comfort and ease of use,3 and offers full integration with our EnSite Precision™ Cardiac Mapping System.
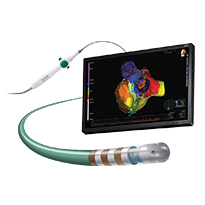
FlexAbility Ablation Catheter, Sensor Enabled
Designed to enhance procedural versatility and precision when integrated with Abbott mapping and navigation systems, the FlexAbility Ablation Catheter, Sensor Enabled™, was created to facilitate detailed, accurate model creation3 and effective lesion creation.4

This device is commercially available for use in select international markets.
* Faster procedures allows us to treat more patients.
References
- Tilz, R.R. (2025, January 17) Acute results demonstrate safety and effectiveness of balloon-based pulsed field ablation system for de novo PVI in PAF and PersAF [Late Breaking Presentation]. AF Symposium 2025, Boston MA, USA.
- Nair D et al. Acute Results of a Novel Flexible Tip Radiofrequency Catheter Incorporating Contact Force Sensing. EP Europace. (In press)
- Abbott. Data on file. Report 90349982.
- Abbott. Data on file. Report 90213470.
MAT-2209121 v2.0