About Coronary Artery Disease (CAD)
Coronary artery disease (CAD) develops when plaque—a combination of cholesterol and other substances—builds up in the walls of the arteries that supply blood to the heart and other parts of the body (called atherosclerosis). This buildup of plaque causes the inside of the arteries to narrow over time, which can partially or totally block blood flow to the heart (called myocardial ischemia).1,2
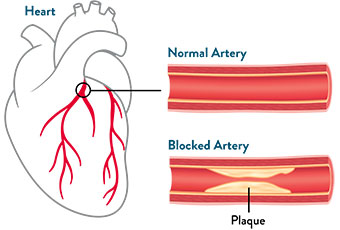
With CAD, the coronary arteries become narrower and harden over many years, reducing oxygen-rich blood flow to the heart.1
About 20.1 million American adults have coronary artery disease, making it the most common type of heart disease in the United States.3
What Are the Symptoms of CAD?
Symptoms of coronary artery disease occur when the heart doesn't get enough oxygen-rich blood.1,2
- Reduced blood flow to the heart can cause chest pain or discomfort (angina) and shortness of breath
- These symptoms may get worse as the buildup of plaque continues to narrow the coronary arteries
- A complete blockage of blood flow can cause a heart attack. Symptoms of heart attack include:
- Angina
- Weakness, light-headedness, nausea (feeling sick to your stomach), or a cold sweat
- Pain or discomfort in the arms or shoulder
- Shortness of breath
A diagnosis of CAD is based on symptoms, medical and family history, risk factors, and the results from tests and procedures.1
CAD can be a silent condition, meaning you may not have any symptoms until you experience a heart attack or other complication.1,2
What Are the Risk Factors for CAD?
Risk factors for coronary artery disease include:1,2
- High blood cholesterol
- High blood pressure
- Family history of CAD
- Diabetes
- Smoking
- Age: being postmenopausal for women and being older than 45 for men
- Obesity
How Is CAD Treated?
If you have been diagnosed with coronary artery disease, your treatment will depend on how serious your symptoms are. To help lower your risk for heart attack or worsening heart disease, treatment may include:1,2
- Heart-healthy lifestyle changes, such as eating a healthier (lower sodium, lower fat) diet, increasing physical activity, reaching a healthy weight, and quitting smoking
- Medicines to treat risk factors for CAD, such as high cholesterol, high blood pressure, and diabetes
- A procedure or heart surgery to help restore blood flow to the heart
- Percutaneous coronary intervention (PCI) is a common method to open coronary arteries that are narrowed or blocked by the buildup of plaque. Blood flow is restored using a variety of medical devices. Many people require a stent (small mesh-like tube device) to keep the coronary arteries open once blood flow has been restored
- Coronary artery bypass grafting (CABG) improves blood flow to the heart by using normal arteries from the chest wall and veins from the legs to bypass the blocked arteries. Surgeons typically use CABG to treat people who have severe coronary artery disease in multiple coronary arteries
References
- National Heart Lung and Blood Institute website. Coronary heart disease. https://www.nhlbi.nih.gov/health/coronary-heart-disease. Accessed October 24, 2022.
- Centers for Disease Control and Prevention website. Coronary artery disease (CAD). https://www.cdc.gov/heartdisease/coronary_ad.htm Accessed October 24, 2022.
- Heart Disease and Stroke Statistics-2022 Update: A Report From the American Heart Association (v1.0) for updated CHD in US (20.1 million)
MAT-2205023 v2.0
