Accessories
AMG PMP Adult Oxygenator
The PMP Adult Oxygenator designed from the ground up for hemocompatibility.

A high flow oxygenator (7.0 L/min) with low priming volume (220 ml), the AMG PMP Adult Oxygenator was built for excellent gas transfer and minimal blood stagnation.
Manufactured by Eurosets and distributed by Abbott, the AMG PMP Adult Oxygenator easily integrates with the CentriMag™ System for ECMO support for up to 14 days. Similar to the CentriMag System, the AMG PMP Adult Oxgyenator maximizes gentle blood handling in the circuit.
The AMG PMP Adult Oxygenator features:
- PMP fibers – for reliable gas exchange performance over duration of use
- PC Coating – to minimize inflammation1 and thrombosis2
- Stainless steel heat exchanger – can eliminate risk of any potential contamination
- Low shear stress – based on shape, fiber orientation, and blood flow path
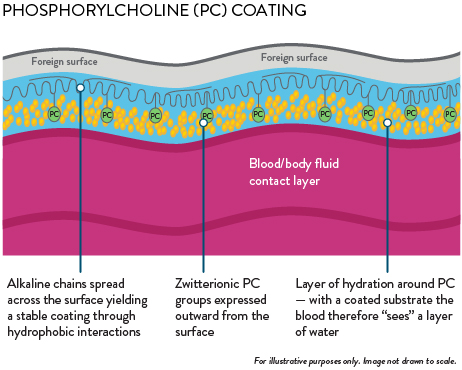
Blood path fully coated with phosphorylcholine (PC) coating
PC creates a permanent water barrier between blood and the oxygenator surface that minimizes thrombus formation and promotes resistance to bacterial adhesion.3
Integrated heat-exchanger maximizes heat exchange efficiency
The integrated heat exchanger is:
- Made of stainless steel for high heat conductivity
- Non-porous to eliminate risk of bacterial infection or any other potential contamination
- Corrugated to increase surface area for efficient heat exchange

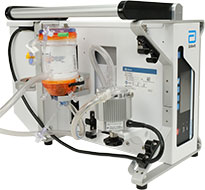
CentriMag System Transporter
The CentriMag System is fully transportable via air or ground ambulance using the compact system transporter, which features:
- Space for core system components, including console, pump/motor and oxygenator
- No ground plate required; CentriMag Pump impeller maintains its orientation automatically through Full MagLev™ Flow Technology
- Machined aluminum oxygenator bracket, compatible with all Eurosets AMG PMP Oxygenators and the CentriMag Adult Oxygenator Kit
- Tested against 60601-1-12 EMS standard for air and ground transportation
- Collapsible hooks on back of transporter allow frame to mount onto rail
This device is commercially available for use in select international markets.
AMG PMP Oxygenator and Oxygenator Holder manufactured by Eurosets s.r.l., and distributed by Abbott.
*PMA approval for 30-day use of CentriMag™ System components include: CentriMag™ Pump, CentriMag™ Console, CentriMag™ Motor, Mag Monitor, flow probe, and CentriMag™ Drainage Cannula and CentriMag™ Return Cannula. Optional accessories include: CentriMag™ System Cart, CentriMag™ System Transporter and Pressure Transducer. PMA approval for 30-day use of CentriMag™ System excludes: PediMag™ Blood Pump and any other pediatric components or accessories.
References
- Corno A, Faulkner G, Harvey C. Mobile extracorporeal membrane oxygenation. ASAIO Journal. 2020. doi:10.1097/MAT.00000000000012786
MAT-2409114 v1.0
