Introducing the UltiPace Pacing Lead, newly engineered from Abbott, the first and only provider of stylet-driven leads that are FDA approved for left bundle branch area pacing (LBBAP).
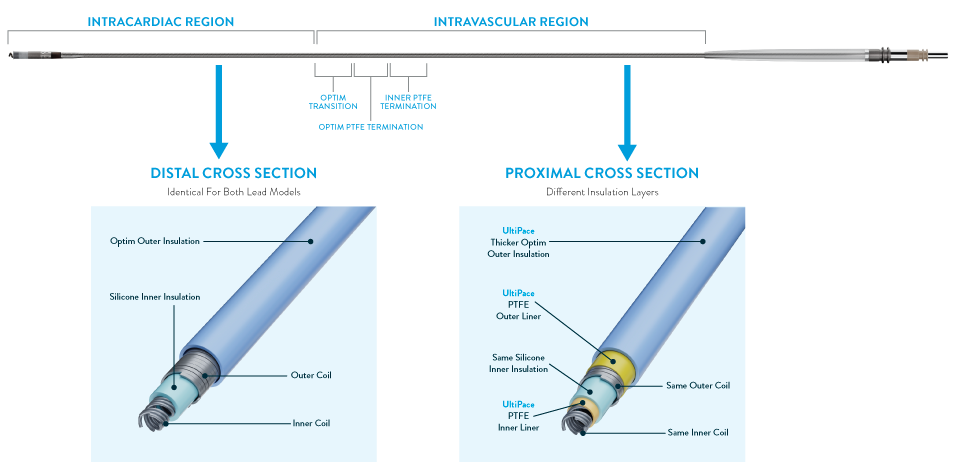
UltiPace Pacing Leads carry forward the proven distal design of Tendril STS while the proximal lead end has been enhanced with additional layers of insulation to provide increased abrasion and crush resistance.1
Increase in abrasion resistance
Increase in lead crush resistance
Increase in abrasion resistance
Increase in lead crush resistance
COMPOSITE
SUCCESS
RATE2*
of patients not experiencing
LBBAP-related adverse effects at 6 months post-implant.2

UltiPace Pacing Leads have a robust distal lead tip without a flexible zone that helps reduce stress on the conductors and the chance for distal fatigue failure compared to competitive leads.3

The Helix Locking Tool is a proven solution to lock an extended helix during tissue burrowing without retraction.3

Improvement in torsional stiffness when using UltiPace Pacing Leads compared to LBBAP-approved lumenless pacing leads.3
Provides the user with uninterrupted feedback to help achieve optimal results.4

References
*Composite success rate of acceptable capture thresholds and sense amplitudes for LBBAP at 6 months post-implant.
Indications:
UltiPace™ leads are indicated for use in combination with a compatible pacemakers, implantable cardioverter defibrillator (ICDs) or cardiac resynchronization therapy (CRT-P/CRT-D) to provide sensing and pacing for the management of chronic symptomatic bradycardia and various atrioventricular conduction abnormalities in patients who experience syncope, presyncope, fatigue, disorientation due to arrhythmia/ bradycardia, or any combination of these symptoms. The UltiPace leads are implanted transvenously in either the right atrium, the right ventricle or the left bundle branch area.
MAT-2403244 v2.0
Stay Connected